May
30
Penn State scientists have found that coal may represent a potential way to store hydrogen gas, much like batteries store energy for future use, addressing a major hurdle in developing a clean energy supply chain. The potential solution coal offers to store hydrogen could bring a hydrogen economy closer to practicality.
The report of the findings has been published in the journal Applied Energy.
Shimin Liu, associate professor of energy and mineral engineering at Penn State said, “We found that coal can be this geological hydrogen battery. You could inject and store the hydrogen energy and have it there when you need to use it.”
Hydrogen is a clean burning fuel and shows promise for use in the most energy intensive sectors of our economy – transportation, electricity generation and manufacturing. But much work remains to build a hydrogen infrastructure and make it an affordable and reliable energy source, the scientists noted.
This includes developing a way to store hydrogen, which is currently expensive and inefficient. Geologic formations are an intriguing option, the scientists said, because they can store large amounts of hydrogen to meet the peaks and valleys as energy demand changes daily or seasonally.
Liu pointed out, “Coal is well-studied, and we have been commercially producing gas from coal for almost a half century. We understand it. We have the infrastructure. I think coal would be the logical place to do geological hydrogen storage.”
To put this to the test, the scientists analyzed eight types of coals from coalfields across the United States to better understand their sorption and diffusion potential, or how much hydrogen they can hold.
All eight coals showed considerable sorption properties, with low-volatile bituminous coal from eastern Virgina and anthracite coal from eastern Pennsylvania performing the best in tests, the scientists reported in Applied Energy.
“I think it’s highly possible that coal could be the very top selection for geological storage from a scientific perspective,” said Liu. “We find that coal outperforms other formations because it can hold more, it has existing infrastructure and is widely available across the country and near populated areas.”
Depleted coalbed methane reservoirs may be the best candidates. These seams contain unconventional natural gas like methane and have become an important source of fossil fuel energy over the last several decades. The methane sticks to the surface of the coal, in a process called adsorption.
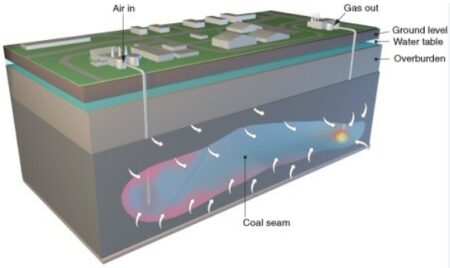
Underground Coal Gasification. Image Credit: Science & Technology Review. From the site Underground Coal Gasification, DOE National Energy Technology Laboratory.
Similarly, injecting hydrogen into coal would cause that gas to absorb or stick to the coal. These formations often have a layer of shale or mudstone on top that act as a seal keeping methane, or in this case hydrogen, sealed until it is needed and pumped back out, the scientists said.
“A lot of people define coal as a rock, but it’s really a polymer,” Liu said. “It has high carbon content with a lot of small pores that can store much more gas. So coal is like a sponge that can hold many more hydrogen molecules compared to other non-carbon materials.”
The scientists designed special equipment to conduct the experiments. Coal has a weaker affinity with hydrogen compared to other sorbing gases like methane and carbon dioxide, so traditional pressurized equipment for determining sorption would not have worked.
“We did a very novel and very challenging design,” Liu said. “It took years to figure out how to do this properly. We had to properly design an experiment system, trial and error based on our previous experience with coals and shales.”
Based on their results, the scientists determined anthracite and semi-anthracite coals are good candidates for hydrogen storage in depleted coal seams, and low-volatile bituminous coal are better candidates for gassy coal seams.
Developing hydrogen storage in coal mining communities could bring new economic opportunities to these regions while also helping create the nation’s hydrogen infrastructure.
“In the energy transition, it’s really coal communities that have been the most impacted economically,” Liu said. “This is certainly an opportunity to repurpose the coal region. They already have the expertise – the energy engineer and skills. If we can build an infrastructure and change their economic opportunities – I think that’s something we should consider.”
Future work will focus on the dynamic diffusivity and dynamic permeability of coal, features which determine how quickly hydrogen can be injected and pumped back out, the scientists said.
“I think Penn State is the right place to do all this research – we have the coal reserves, we have natural gas, we have both the engineering and economic expertise at the University,” Liu said. “This is the logical place to do this.”
Also contributing from Penn State was Ang Liu, instructor, John and Willie Leone Family Department of Energy and Mineral Engineering.
***
Your humble author heartily agrees. Hydrogen is the smallest atom. H2 or dihydrogen is the smallest molecule. Its simply doesn’t stay put. Gravity is so far as known today – the best containment device. And the earth’s gravity isn’t really strong enough.
There remain some early questions. Foremost is the amount of hydrogen needed to get the coal saturated enough to give back what is put in. Next would be the rate the hydrogen escapes. Then the reactive loss to the coal and the products that result.
Just knowing that the hydrogen sticks and stays put a while is a huge breakthrough. How this technology develops could be very significant in the hydrogen economy’s growth.
There could be competition from other polymers. We might learn how to make safe low cost storage for business home and transport use.
For hydrogen, this is a milestone marker of considerable significance.
Thanks and congratulations are in order for this team. One hopes everyone sees the significance of this work.

