Mar
19
New Fuel Cell Design Makes Double the Voltage
March 19, 2019 | 2 Comments
Washington University in St. Louis engineers have developed a high-powered fuel cell that operates at double the voltage of today’s commercial fuel cells. It could power underwater vehicles, drones and eventually electric aircraft at a significantly lower cost.
The transportation industry is one of the largest consumers of energy in the U.S. economy with increasing demand to make it cleaner and more efficient. While more people are using electric cars, designing electric-powered planes, ships and submarines is much harder due to power and energy requirements.
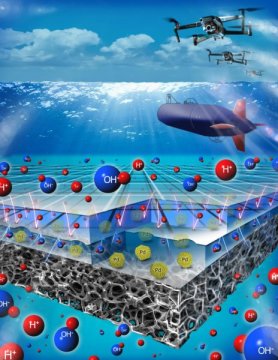
This is an artistic representation of the pH-gradient enabled microscale bipolar interface (PMBI) created by Vijay Ramani and his lab. The two layers that make up the interface are covering the third bottom layer, which is the electrode with palladium particles on it. The submarine and drones are envisioned applications of the direct borohydride fuel cell which incorporates the PMBI. Image Credit: McKelvey School of Engineering, Washington University. Click image for the largest view.
A team of engineers in the McKelvey School of Engineering at Washington University in St. Louis has developed a high-power fuel cell that advances technology in this area. Led by Vijay Ramani, the Roma B. and Raymond H. Wittcoff Distinguished University Professor, the team has developed a direct borohydride fuel cell that operates at double the voltage of today’s commercial fuel cells.
The paper detailing the advancement using a unique pH-gradient-enabled microscale bipolar interface (PMBI), has been published in Nature Energy. The improvement could power a variety of transportation modes – including unmanned underwater vehicles, drones and eventually electric aircraft – at significantly lower cost.
Ramani, also professor of energy, environmental & chemical engineering said, “The pH-gradient-enabled microscale bipolar interface is at the heart of this technology. It allows us to run this fuel cell with liquid reactants and products in submersibles, in which neutral buoyancy is critical, while also letting us apply it in higher-power applications such as drone flight.”
The fuel cell uses an acidic electrolyte at one electrode and an alkaline electrolyte at the other electrode. Typically, the acid and alkali will quickly react when brought in contact with each other. Ramani said the key breakthrough is the PMBI, which is thinner than a strand of human hair. Using membrane technology developed at the McKelvey Engineering School, the PMBI can keep the acid and alkali from mixing, forming a sharp pH gradient and enabling the successful operation of this system.
Shrihari Sankarasubramanian, a research scientist on Ramani’s team said, “Previous attempts to achieve this kind of acid-alkali separation were not able to synthesize and fully characterize the pH gradient across the PMBI. Using a novel electrode design in conjunction with electroanalytical techniques, we were able to unequivocally show that the acid and alkali remain separated.”
Lead author Zhongyang Wang, a doctoral candidate in Ramani’s lab, added, “Once the PBMI synthesized using our novel membranes was proven to work effectively, we optimized the fuel cell device and identified the best operating conditions to achieve a high-performance fuel cell. It has been a tremendously challenging and rewarding pathway to developing the new ion-exchange membranes that has enabled the PMBI.”
“This is a very promising technology, and we are now ready to move on to scaling it up for applications in both submersibles and drones,” Ramani said.
Other participants in this work include Cheng He, a doctoral candidate, and Javier Parrondo, a former research scientist in Ramani’s lab. The team is working with the university’s Office of Technology Management to explore commercialization opportunities.
Double the voltage is always a good thing at the levels where fuel cells and batteries work. Cars, trucks, ships and trains are good prospects, but flying might prove more of a challenge. But never say never, the past decade here has been both a surprise and disappointment on what comes to market.
Comments
2 Comments so far


This is a new thought. If it is widely used, the environment will be better.
Technology has been developing and we need to pay attention to the environment.