Oct
18
Another New Solar Photocatalyst For Making Hydrogen Fuel
October 18, 2018 | Leave a Comment
Daegu Gyeongbuk Institute of Science and Technology (DGIST) researchers led by DGIST Professor Jong-Sung Yu’s team at the Department of Energy Science and Engineering have developed new photocatalyst synthesis method using Magnesium hydride (MgH2) and Titanium dioxide (TiO2). The result is expected to contribute to hydrogen mass production through the development of photocatalyst that reacts to solar light.
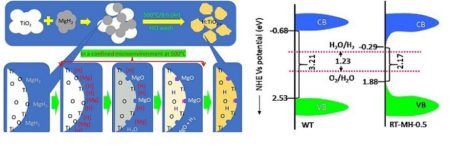
This diagram illustrates the surface reactions and characteristics of reduced titanium based photocatalysts developed at DGIST. Image Credit: DGIST. Click image for the largest view.
Intense research is being conducted globally on how to produce hydrogen using solar light and photocatalysts for decomposing water. To overcome the limitations of photocatalysts that only reacts to light in ultraviolet rays, researchers have doped dual atom such as Nitrogen (N), Sulfur (S), and Phosphorus (P) on photocatalyst or synthesized new photocatalysts, developing a photocatalyst that reacts efficiently to visible light.
The team detailed their work in a paper published in the journal Applied Catalysis B: Environmental.
With Professor Samuel Mao’s team at UC Berkeley in the U.S., Professor Yu’s research team developed a new H-doped photocatalyst by removing oxygen from the photocatalyst surface made of titanium dioxide and filling hydrogen into it through the decomposition of MgH2. Energy of long wavelength including visible light could not be used for the existing white Titanium dioxide because it has a wide band gap energy. However, the development of MgH2 reduction could overcome this through oxygen flaw induction and H-doping while enabling the use of solar light with 570nm-wavelength.
MgH2 reduction can synthesize new materials by applying to Titanium oxide used in this research as well as the oxides composed of other atoms such as Zr, Zn, and Fe. This method is applicable to various other fields such as photocatalyst and secondary battery. The photocatalyst synthesized in this research has four times higher photoactivity than the existing white titanium dioxide and is not difficult to manufacture, thus being very advantageous for hydrogen mass production.
Another characteristic of the photocatalyst developed by the research team is that it reduces band gap more than the existing Titanium dioxide photocatalyst used for hydrogen generation and can maintain four times higher activity with stability for over 70 days.
The new method can also react to visible light unlike existing photosynthesis, overcoming the limitation of hydrogen production. With the new photocatalyst development, the efficiency and stability of hydrogen production can both dramatically improved, which will help popularize hydrogen energy in the near future.
Professor Yu said, “The photocatalyst developed this time is a synthesis method with much better performance than the existing photocatalyst method used to produce hydrogen. It is a very simple method that will greatly help commercialize hydrogen energy. With a follow-up research on improving the efficiency and economic feasibility of photocatalyst, we will take the lead in creating an environment stable hydrogen energy production that can replace fossil energy.”
The hydrogen fuel photocatalysts are popping up quite often lately. Its seems that one or more could have real potential. This one seems to use low cost materials and lasts a fairly long while, two important attributes.
There is still quite a way to go for roof top hydrogen production. Any and all of what we’ve been seeing have to be proven up, packaging developed for outdoor exposure and a long list of the things about getting to market. Then the real bugaboos get underway.
That’s, storage, safety and something to use the fuel. We’re not seeing those products offered at mass scale at all so far.

