Oct
31
Defects & Disordered Material Make Better Lithium Batteries
October 31, 2017 | 4 Comments
Rice University scientists are using defects for high performance lithium ion batteries while Berkeley Lab scientists can make up disordered materials for better lithium ion cathodes.
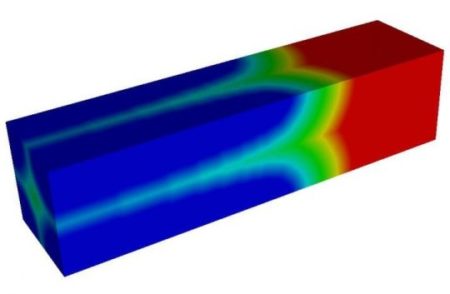
An illustration shows the growth of a lithium-deficient phase (blue) at the expense of a Lithium-rich phase (red) in a lithium iron phosphate microrod. Image Credit: Mesoscale Materials Modeling Group/Rice University. Click image for the largest view.
Rice materials scientist Ming Tang and chemists Song Jin at the University of Wisconsin-Madison and Linsen Li at Wisconsin and the Massachusetts Institute of Technology led a study that combined state-of-the-art, in situ X-ray spectroscopy and modeling to gain insight into lithium transport in battery cathodes. They found that a common cathode material for lithium-ion batteries, olivine lithium iron phosphate, releases or takes in lithium ions through a much larger surface area than previously thought.
Tang said, “We know this material works very well but there’s still much debate about why. In many aspects, this material isn’t supposed to be so good, but somehow it exceeds people’s expectations.”
That and other revelations appear in paper published in Nature Communications that could potentially help manufacturers develop better lithium-ion batteries that power electronic devices worldwide.
The lead authors of the study – Liang Hong of Rice and Li of Wisconsin and MIT – and their colleagues collaborated with Department of Energy scientists at Brookhaven National Laboratory to use its powerful synchrotron light sources and observe in real time what happens inside the battery material when it is being charged. They also employed computer simulations to explain their observations.
One revelation, Tang said, was that microscopic defects in electrodes are a feature, not a bug, “People usually think defects are a bad thing for battery materials, that they destroy properties and performance. With the increasing amount of evidence, we realized that having a suitable amount of point defects can actually be a good thing.”
Inside a defect-free, perfect crystal lattice of a lithium iron phosphate cathode, lithium can only move in one direction, Tang explained. Because of this, it is believed the lithium intercalation reaction can happen over only a fraction of the particle’s surface area.
But the team made a surprising discovery when analyzing Li’s X-ray spectroscopic images: The surface reaction takes place on the large side of his imperfect, synthesized microrods, which counters theoretical predictions that the sides would be inactive because they are parallel to the perceived movement of lithium.
The researchers explained that particle defects fundamentally change the electrode’s lithium transport properties and enable lithium to hop inside the cathode along more than one direction. That increases the reactive surface area and allows for more efficient exchange of lithium ions between the cathode and electrolyte.
Because the cathode in this study was made by a typical synthesis method, Tang said, the finding is highly relevant to practical applications.
Tang summarized with, “What we learned changes the thinking on how the shape of lithium iron phosphate particles should be optimized. Assuming one-dimensional lithium movement, people tend to believe the ideal particle shape should be a thin plate because it reduces the distance lithium needs to travel in that direction and maximizes the reactive surface area at the same time. But as we now know that lithium can move in multiple directions, thanks to defects, the design criteria to maximize performance will certainly look quite different.”
The second surprising observation, Tang said, has to do with the movement of phase boundaries in the cathode as it is charged and discharged, “When you take heat out of water, it turns into ice. And when you take lithium out of these particles, it forms a different lithium-poor phase, like ice, that coexists with the initial lithium-rich phase.” The phases are separated by an interface, or a phase boundary. How fast the lithium can be extracted depends on how fast the phase boundary moves across a particle.
Unlike in bulk materials, Tang explained, it has been predicted that phase boundary movement in small battery particles can be limited by the surface reaction rate. The researchers were able to provide the first concrete evidence for this surface reaction-controlled mechanism, but with a twist.
Tang explored the research results with, “We see the phase boundary move in two different directions through two different mechanisms, either controlled by surface reaction or lithium bulk diffusion. This hybrid mechanism paints a more complicated picture about how phase transformation happens in battery materials. Because it can take place in a large group of electrode materials, this discovery is fundamental for understanding battery performance and highlights the importance of improving the surface reaction rate.”
The Rice press release sets up the Berkeley Lab release nicely.
Berkeley Lab scientists report major progress in cathodes made with so-called “disordered” materials, a promising new type of lithium battery.
In a pair of papers published this month in Nature Communications and Physical Review Letters (PRL), a team of scientists led by Gerbrand Ceder has come up with a set of rules for making new disordered materials, a process that had previously been driven by trial-and-error. They also found a way to incorporate fluorine, which makes the material both more stable and have higher capacity.
Ceder, a Senior Faculty Scientist at Berkeley Lab who also has an appointment at UC Berkeley’s Department of Materials Science and Engineering overlooked the situation with, “This really seems to be an interesting new direction for making high energy density cathodes. It’s remarkable that all the disordered rock salts people have come up with so far have very high battery capacity. In the PRL paper we give a guideline for how to more systematically make these materials.”
As the Rice paper noted, the cathode material in lithium batteries is typically “ordered,” meaning the lithium and transition metal atoms are arranged in neat layers, allowing lithium to move in and out of the layers.
A few years ago, Ceder’s group discovered that certain types of disordered material could store even more lithium, giving batteries higher capacity.
The Physical Review Letters lead author, Alexander Urban, a Berkeley Lab postdoctoral fellow said, “Despite their attractive properties, discovering new disordered materials has been mostly driven by trial-and-error and by relying on human intuition. Now we have for the first time identified a simple design criterion to predict novel disordered compositions. The new understanding establishes a relationship between the chemical species, local distortions of the crystal structure, and the tendency to form disordered phases.”
The other advantage of using disordered materials is the ability to avoid the use of cobalt, a limited resource, with more than half the world’s supply existing in politically unstable countries. By moving to disordered rock salts, battery designers could be free to use a wider range of chemistries. For example, disordered materials have been made using chromium, titanium, and molybdenum.
Ceder said, “We want the ability to have more compositional freedom, so we can tune other parameters. There are so many properties to optimize – the voltage, the long-term stability, whether it’s easy to synthesize – there’s so much that goes into taking a battery material to a commercial stage. Now we have a recipe for how to make these materials.”
The Berkeley Lab folks popped another connecting topic in their press release.
Another major advance in lithium-ion batteries is reported in the Nature Communications paper, “Mitigating oxygen loss to improve the cycling performance of high capacity cation-disordered cathode materials,” which shows that disordered materials can be fluorinated, unlike other battery materials. Fluorination confers two advantages: it allows more capacity and makes the material more stable. In a battery, the increased stability would translate into a device with long cycle life and that is less likely to catch fire.
The lead author of the paper, Jinhyuk Lee, formerly a Berkeley Lab researcher, worked with scientists at Berkeley Lab’s Advanced Light Source (ALS), a source of X-ray beams for scientific research, to conduct in situ experiments.
Ceder said, “The ALS was really important to understand the mechanism by which we get higher capacity. What’s really cool is you can look at the battery while it’s operating, and look at the electronic structure of the cathodes. So you learn how it charges and discharges, where the electrons go, which is a crucial aspect of charge storage.”
ALS scientists Shawn Sallis and Wanli Yang are co-authors, as is Bryan McCloskey of Berkeley Lab. “His group was crucial in showing these materials are more stable and don’t lose oxygen,” Ceder said.
Now that they have demonstrated the concept, Ceder plans to follow up by trying to add even more fluorine to the materials.
“New cathode materials is the hottest direction in Li-ion batteries,” Ceder said. “The field is a bit stuck. To get more improvements in energy storage there are only a few directions to go. One is solid-state batteries, and the other is to keep improving the energy density of electrode materials. The two are not mutually exclusive. This research line is definitely not exhausted yet.”
This is all superb news, if a little much for a single post. Still all three topics covered are closely related and showed up within hours of one another.
The main issue is manufacturers are seeing developments far faster than they can adapt. Commercial scale has reality demands of economics, investment and certainty of customer acceptance. There is hardly time between research announcements to have a commercial scale investigation of applicability. The good news is some research is going to go to market and the bad news is some of it isn’t. Lets hope the industrial due diligence efforts find the optimal ways to progress.
Comments
4 Comments so far


Still all three topics covered are closely related and showed up within hours of one another.
Commercial scale has reality demands of economics, investment and certainty of customer acceptance. Interesting.
We know this material works very well but there’s still much debate about why. In many aspects, this material isn’t supposed to be so good, but somehow it exceeds people’s expectations
This is actually quite brilliant. We fight entropy but just a little bit, enough for an organic side-effect is often all we need for spectacular, emerging results. This type of paper really reminds me of Wolfram’s computational theories. From the article, “We see a phase boundary move in two different directions through two different mechanisms”. Very interesting indeed. It makes me quite certain the best batteries will be designed much in the way of the ‘game of life’ rather than the entropy-restricted crystalline lattice structures that logic-based labs are producing today.