Jun
9
Breakthrough For Calcium Ion Batteries
June 9, 2016 | Leave a Comment
Calcium ion battery chemistry with double the capacity of lithium ion now has a potential battery electrode solution. (Link is to a pdf download.) Toyohashi University of Technology (TUT) researchers have demonstrated calcium ion batteries (CIBs) using pigment based electrodes such as Prussian Blue and its analogues.
The CIBs showed excellent cyclability of discharge and charge in a calcium-based organic electrolyte. The researchers are thinking the success derives from strong atomic bonds in Prussian blue structures, which possess movable pathways for large-sized ions in three dimensions. Such unique structures have excellent potential for application in newly designed batteries.
The team’s research paper has been published in the journal Electrochimica Acta behind a paywall.
CIBs have attracted much attention as next-generation batteries to replace lithium ion batteries (LIBs) because the theoretical capacity of CIBs is twice that of the LIBs. This doubled capacity can be explained by the difference between monovalent and divalent ions. In addition, CIBs possess advantages such as a lower cost and higher safety because calcium is far more abundant than lithium and because CIBs have a higher melting point than LIBs.
So far there is the one major obstacle to the application in CIBs. There is no suitable electrode material in which calcium ions can be inserted and extracted reversibly because of the relatively large ionic radius of calcium ions (112 pm) as compared to that of lithium ions (76 pm).
For this study Tomohiro Tojo and his colleagues at the Department of Electrical and Electronic Information Engineering of TUT, employed Prussian blue (PB) and Prussian blue analogues (PBAs) as CIB electrodes because they possess large sites for inserting and extracting large-sized ions, as shown in Fig. 1. above. Up to now, using PBAs as an electrode material, the electrochemical behaviors of sodium ions, corresponding to the radius of calcium ions, have been examined in organic and inorganic electrolytes. These reports have shown reversible insertion and extraction of sodium ions into and from PBA structures.
The research team investigated the electrochemical performance of several PBA electrodes in order to determine whether calcium ions in an organic electrolyte exhibit either reversible or irreversible insertion and extraction into and from the crystal structure. The team found reversible capacities of 40-50 mAh/g at a low current density, as shown in Fig. 2(a). In Fig. 2(b), the Coulombic efficiencies, which are defined as the ratio of the amount of insertion (discharge) and extraction (charge) of calcium ions, were observed to have a constant value of 90% after the 3rd cycle.
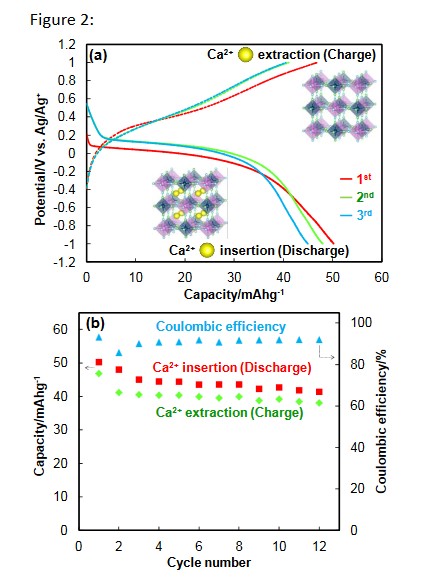
2 (a) Discharge -charge profiles of a Prussian blue analogue electrode in a calcium – based organic electrolyte and 2 (b) cycled capacities with discharge – charge capacity ratio (Coulombic efficiencies).
The results shown in Fig. 2 demonstrate the excellent cyclability of the PBA electrodes, even though the reversible capacities were half the theoretical capacity. The researchers investigated the reason for the high reversibility using X-ray diffraction (XRD) and X-ray photoelectron spectroscopy (XPS). The reversibility is explained by the durable structure of the PBAs and their excellent charge balance during the insertion and extraction of calcium ions.
The team is proposing Prussian blue (PB) and Prussian blue analogues (PBAs) as feasible electrode materials for CIBs, even though additional investigation is needed in order to enhance the reversible capacities still further.
They’re already on it. The researchers have carried out the further study to utilize CIBs beyond LIBs.
Calcium vs. lithium is a very different raw material situation. Lithium is not platinum, but compared to calcium quite expensive. Prussian Blue is a low cost material as well. With this team, and soon to be others, working on it we’ll soon see something like half the weight or twice the power kinds of solutions. For cell phone its a big deal in prices, for electric vehicles, a truly disruptive technology with a very positive impact.


