Aug
12
New Catalyst Makes Carbon Dioxide Into Methanol Fuel
August 12, 2015 | Leave a Comment
Argonne National Laboratory scientists have identified a new material to make methanol from carbon dioxide. With its unique structure, the new catalyst can capture and convert carbon dioxide in a way that ultimately saves energy.
The Argonne scientists are working on the ‘capture and convert’ motto of carbon dioxide reduction, a process that stops the greenhouse gas before it escapes from chimneys and power plants into the atmosphere and instead turns it into a useful product. That would be a huge improvement on the old idea of trying to pump the CO2 back deep underground.
One possible end product is methanol, a liquid fuel and the focus of a recent study published in the Journal of the American Chemical Society and conducted at the U.S. Department of Energy’s (DOE) Argonne National Laboratory. The chemical reactions that make methanol from carbon dioxide rely on a catalyst to speed up the conversion, and Argonne scientists identified a new material that could fill this role. With its unique structure, this catalyst can capture and convert carbon dioxide in a way that ultimately saves energy.
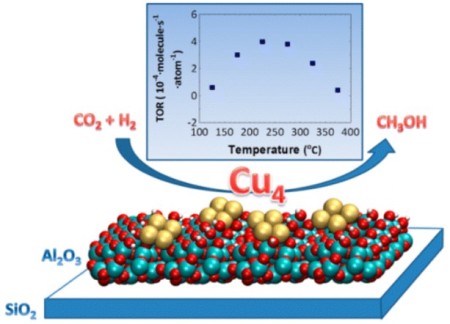
The use of size-selected Cu4 clusters supported on Al2O3 thin films for CO2 reduction in the presence of hydrogen. Image Credit: Argonne National Laboratory. Click image for the largest view.
The Argonne team is calling it a copper tetramer.
It consists of small clusters of four copper atoms each, supported on a thin film of aluminum oxide. The catalyst works by binding to carbon dioxide molecules, orienting them in a way that is ideal for chemical reactions. The structure of the copper tetramer is such that most of its binding sites are open, which means it can attach more strongly to carbon dioxide and can better accelerate the conversion.
The current industrial process to reduce carbon dioxide to methanol uses a catalyst of copper, zinc oxide and aluminum oxide. A number of its binding sites are occupied merely in holding the compound together, which limits how many atoms can catch and hold carbon dioxide.
Stefan Vajda, senior chemist at Argonne and the Institute for Molecular Engineering and co-author on the paper said, “With our catalyst, there is no inside. All four copper atoms are participating because with only a few of them in the cluster, they are all exposed and able to bind.”
To compensate for a catalyst with fewer binding sites, the current method of reduction creates high-pressure conditions to facilitate stronger bonds with carbon dioxide molecules. But compressing the gas into a high-pressure mixture takes a lot of energy.
The benefit of enhanced binding is that the new catalyst requires lower pressure and less energy to produce the same amount of methanol.
Carbon dioxide emissions are believed by some to be an ongoing environmental problem, and according to the authors, it’s important that research identifies optimal ways to deal with the waste.
Larry Curtiss, an Argonne Distinguished Fellow who co-authored the paper said, “We’re interested in finding new catalytic reactions that will be more efficient than the current catalysts, especially in terms of saving energy.”
Copper tetramers could allow us to capture and convert carbon dioxide on a larger scale – reducing a potential environmental threat and creating a useful product like methanol that can be transported and used for fuel.
The new catalyst still has a long journey ahead from the lab to industry. Potential obstacles include instability and figuring out how to manufacture mass quantities. There’s a chance that copper tetramers may decompose when put to use in an industrial setting, so ensuring long-term durability is a critical step for future research, Curtiss said. And while the scientists needed only nanograms of the material for this study, that number would have to be multiplied dramatically for industrial purposes.
Meanwhile, the researchers are interested in searching for other catalysts that might even outperform their copper tetramer.
These catalysts can be varied in size, composition and support material, which results in a list of more than 2,000 potential combinations, Vajda said.
But the scientists don’t have to run thousands of different experiments, said Peter Zapol, an Argonne physicist and co-author of this paper. Instead, they will use advanced calculations to make predictions, and then test the catalysts that seem most promising.
“We haven’t yet found a catalyst better than the copper tetramer, but we hope to,” Vajda said. “With global warming becoming a bigger burden, it’s pressing that we keep trying to turn carbon dioxide emissions back into something useful.”
For this research, the team used the Center for Nanoscale Materials as well as beamline 12-ID-C of the Advanced Photon Source, both DOE Office of Science User Facilities.
Curtiss said the Advanced Photon Source allowed the scientists to observe ultralow loadings of their small clusters, down to a few nanograms, which was a critical piece of this investigation.
The carbon dioxide to methanol path has always been prohibitively expensive to the point of simple impracticality. Perhaps the Argonne team has a breakthrough. It would be welcome. Then we’d get double duty from notorious CO2 emitters like coal. If coal did twice the work for the same effluent, there wouldn’t be so much to complain about.
Like all Argonne press releases, this one was on point and covered the status and obvious forthcoming challenges. But the goal is worthy and lets hope the research continues on.

