Feb
17
Organic Peptides Link Up Lithium Ion Battery Cathodes
February 17, 2015 | 1 Comment
Researchers at the University of Maryland, Baltimore County (UMBC) are using organic peptides to improve the properties of lithium ion batteries. Peptides are molecules of amino acids that are a type of biological molecule. The UMBC team has found one that binds strongly to lithium manganese nickel oxide (LMNO), a material that can be used to make the cathode in high performance lithium ion batteries.
The peptide attaches to nanosized particles of LMNO and connects them to conductive components of a battery electrode. The connection improves the potential power and stability of the electrode.
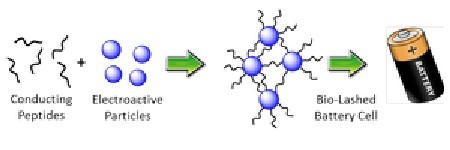
In this schematic, bifunctional peptides that bind to conducting nanotubes are allowed to bind to electroactive particles. The peptides lash together conductor with electroactive particles to make bio-lashed battery electrodes. Image Credit: Mark Allen Lab, UMBC.
Evgenia Barannikova, a graduate student at UMBC who works in the lab of Mark Allen and studies how biological molecules in general can improve the properties of inorganic materials in batteries explains the background, “Biology provides several tools for us to solve important problems. By mimicking biological processes we can find the better solution.”
One of the problems currently facing battery researchers is the difficulty of working with nanoscale materials due to their extra tiny size making them hard to control and hold in place.
The frustrations of working with nanosized materials are worth overcoming, however. Nanostructured electrodes in Li-ion batteries have several advantages over bulk material electrodes, including shorter distances for charge-carrying particles to travel and a high surface area that provides more active sites for electrochemical reactions to occur, all of which translates to batteries that are lighter and longer-lasting.
To take on the challenge of manufacturing on the nanoscale Barannikova and her colleagues turned for help from the biological molecules called peptides. Peptides are made up from strings of molecules known as amino acids. Peptides are naturally occurring and bind to many different types of organic and inorganic materials, depending on their sequence of the amino acids. They play many roles in the human body, from signaling in the brain to regulating blood sugar, and some drugs, like insulin, are made up of peptides.
Barannikova said one of the inspirations for her research was the way that organisms such as mollusks use peptides to control the growth of their shells noting they demonstrate remarkable control in order to build intricate nano- and macrostructures from inorganic materials like calcium carbonate.
The researchers borrowed the general approach of the mollusks, but had to employ some lab-bench wizardry to find the appropriate peptide. No snail, after all, makes its shell from lithium manganese nickel oxide.
Barannikova and her colleagues used a procedure called “Phage Display” to screen more than one billion possible peptides in search of one that would stick strongly to lithium manganese nickel oxide. The “peptide library” through which the researchers searched is commercially produced by a laboratory supply company, and contains a vast number of randomly combined amino acid sequences incorporated into a protein made by a virus called the M13 bacteriophage.
The research team isolated a peptide that binds to lithium manganese nickel oxide by combining the library with a sample of the metal oxide and then repeatedly washing away the peptides that didn’t stick to it. The researchers then combined the newly-discovered peptide with a previously isolated peptide that binds to carbon nanotubes. Carbon nanotubes can serve as conductive nanowires in Li-ion electrodes.
The resulting peptide could then form a bridge, binding to both the lithium manganese nickel oxide nanoparticles and the carbon nanotubes and keeping them close to each other so that they can maintain a connection through multiple charging cycles. By helping to maintain a highly organized architecture at the nanoscale, the researchers expect that their peptides will improve the power and cycling stability of future Li-ion batteries, allowing them to be smaller and maintain longer lifetimes.
The team is currently testing how well the new cathodes perform. Looking forward, Barannikova plans to make an anode with similar techniques and to integrate the two components. “I hope to demonstrate an entire biotemplated battery in my Ph.D. thesis,” she said.
We’re send the strongest of congratulations and wishing the best of luck and support for the continued effort. Should she find a way to economically build nanoscaled battery electrode sets and come up with an electrolyte that enhances, or doesn’t simply erode the peptide connection. the lithium ion battery might see an even lower cost per watt stored.
We might also hope she and her team have a look at the other battery chemistries as the lithium research matures.
Comments
1 Comment so far


I didn’t know that organic peptides could improve potential power and stability of electrodes. Do you have a drawn out mechanism for this reaction? Is it a reaction that has a stable equilibrium?