May
4
A New Break Into Carbon Dioxide Capture
May 4, 2012 | 5 Comments
Texas A&M University chemist Hong-Cai “Joe” Zhou in collaboration with Hae-Kwon Jeong and Perla B. Balbuena in the Department of Chemical Engineering, assemble MOF materials with profound potential for cleaner energy across the globe.
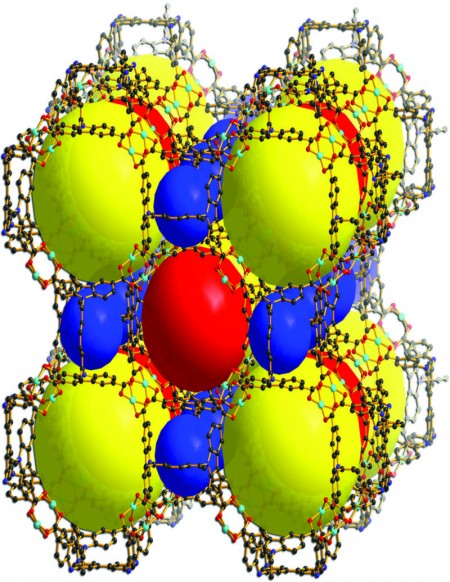
The building blocks the group specializes in actually are a recently developed, increasingly versatile class of materials known as metal-organic frameworks (MOF). An emerging technology in the scientific community, MOFs are porous crystalline polymers made up of metal ions or metal-containing components and organic ligands.
Zhou says, “It’s very fair to say that, in the last decade, the fastest-growing field in chemistry is the study of metal-organic frameworks. The MOF field was formed only about 15 years ago, but it has already shown a lot of promise. We are just one of many teams worldwide working with this exciting new type of material, because the scope of the research is enormous.”
Zhou and his team focus on MOF’s ability to selectively capture carbon dioxide from the exhaust of coal-fired power plants. Though coal is a cheap natural resource, its long-term and widespread use has been a main contributor to the rapidly increasing levels of carbon dioxide in the atmosphere. Zhou notes that capturing carbon dioxide using MOF, coupled with proper sequestration and/or utilization, not only would slow down the escalation of greenhouse gas levels but also allow power plants to continue using inexpensive coal.
Zhou explains that while MOFs come in a huge number of varieties, only a fraction is suitable for carbon capture. Finding that fraction and then maximizing its potential represents the crux of the tedious yet vital chore facing Zhou and his team.
Compounding the complicated matter of piecing together the correct framework is the fact that only a handful of places worldwide conduct large-scale tests on carbon-capture techniques, given the energy industry’s somewhat understandable reluctance to implement such experimental, power-sapping processes. Zhou explains that even the most current state-of-the-art carbon-capture procedure would lead to a 30 percent parasitic power consumption, thereby significantly reducing the power plant’s overall efficiency.
Zhou’s group may have found the alternative. He said they are in the process of constructing a unique subset of MOF that can capture carbon dioxide with extremely high selectivity while using much less power than what is required by commonly applied carbon-capture methods. The group’s goal is to create an MOF that binds only with carbon dioxide and is robust enough to withstand the harsh conditions of the flue gas, resulting in a more economical carbon-capture technique. If successful, it could significantly reduce the amount of carbon dioxide currently being emitted into the atmosphere.
With good progress at hand Zhou readily admits the work with carbon-capturing MOF is far from finished. He says his group’s next big undertaking will be to determine if carbon dioxide can be separated from a flue gas – the exhaust from chimneys, ovens and steam generators – using MOF. In addition, he says there is much more research to be conducted with MOF’s ability to store hydrogen and methane, efforts which will continue into the indefinite future.
The MOF future does look bright, indeed. Zhou said, “In terms of the scope of potential application of MOF, we have barely scratched the surface,” noting beyond carbon capture, MOFs may become useful in gas separation in general.
“Using this new material, the gasses would come in, and the ones that are the right size would stay, while the others would pass. Separation can be performed at a fraction of the original cost using cryo-distillation,” Zhou explains, “Normally in the chemical and petroleum industry, one of the most energy-intensive procedures is the separation of gases, considering you have to liquefy them by compressing and then cooling them. Then you have to do distillation by evaporating and cooling what you wanted to separate. It’s a total waste of energy.”
Beginning in the 1990s, MOFs have been seen with a bright future as an eco-friendly technology that could provide for major improvements in natural gas usage for transportation and in the commercialization of hydrogen-powered vehicles. In their crystalline form, they appear to resemble nothing more than ordinary table salt.
Looks, however, are deceiving, considering MOF have the highest internal surface area known to man. Once unraveled, one sugar-cube-sized piece could cover an entire football field.
In addition to having exceptionally high porosity, Zhou says they are the most tunable material of any known substance. With just a tweak of their crystalline structure and surface properties, they become ideal for absorbing any type of different molecule, lending to their versatility in application.
MOF technology is perhaps still a gestational field. As noted above the possible combinations to make MOFs is astonishing. The ‘break’ Zhou is experiencing is a solid step to the breakthrough. But the work is not just find a set of MOFs that work with CO2 at the situations demand, but the MOF has to be made, commercially, flush freed of the CO2 cheaply and last a long time. Finding that could take a while.
But Zhou is leading the field. His group is zeroing in a on a highly useful application. And the application is soon to be much more important than greenhouse gas when the media and populace realize CO2 is the circulation system of life on earth.
Comments
5 Comments so far


Wow! Very innovative and useful.the commercialization of this will have a huge effect on natural gas production and climatic controls. this could make stripping CO2 much more economical.
Hey very nice website!! Man .. Excellent .. Wonderful .
. I’ll bookmark your web site and take the feeds additionally? I’m glad to seek out so many helpful
information right here within the publish, we
want develop more techniques in this regard, thank you for
sharing. . . . . .
What’s up everyone, it’s my first pay a quick visit
at this web site, and post is really fruitful in support of me, keep
up posting these types of articles.
Yes! Finally something about laptop computers.
Thanks for finally writing about > A New Break Into Carbon
Dioxide Capture | New Energy and Fuel < Liked it!