Apr
28
Titanium Oxide Solar Photocatalysis For Making Fuel
April 28, 2011 | 3 Comments
Solar energy as a power source with its direct conversion into chemical energy, for example hydrogen generation by splitting water, is one of solar energy’s most interesting uses. Titanium dioxide-based photocatalysis are the most efficient today, yet little is understood about solar driven chemical process.
Hydrogen production from water and sunlight by means of oxide powders has been studied extensively for several decades; the basic physical and chemical mechanisms of the processes involved haven’t been described in a satisfactory way so far.
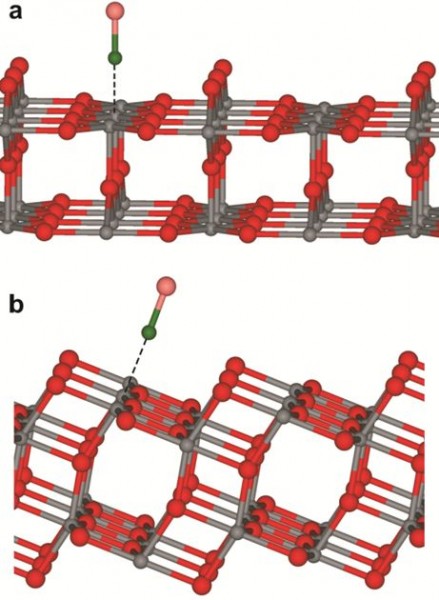
Titanium dioxide, or titania, is a photoactive material that occurs naturally in the rutile and anatase forms. The anatase form is characterized by a desirable ten times higher photochemical activity than the rutile form. When the white TiO2 powder, which is also used as a pigment in paints and sunscreens, is exposed to light, electrons are excited and can; for example, split water into it’s the components of oxygen and hydrogen.
The hydrogen produced in that way is a “clean” energy source: No other chemical products are generated to make controversy and only water is produced during combustion or recombining the hydrogen and oxygen. Titanium dioxide is also used to manufacture self-cleaning surfaces from which unwanted films are removed through photochemical processes triggered by incident sunlight. In hospitals, this effect is used for sterilizing specially coated instruments by means of UV irradiation.
Its relatively common stuff, and the European team is digging in to understand it better and exploit the natural effect.
The titania powder particles used in photoreactors are tiny, only a few nanometers, and are thus too small for use in studies by means of the common and powerful methods of surface analysis. By using instead millimeter-sized single-crystal substrates, the researchers were for the first time able to precisely study photochemical processes on the surface of titanium dioxide by means of a novel infrared spectrometer.
Using a laser-based technique, the scientists determined the lifetime of light-induced electronic excitations inside the TiO2 crystals.
Professor Wöll, who is also head of the IFG, says exact information about these processes is of great importance. He explains the importance of electron excitation lifetime, “A short lifetime means that the excited electrons fall back again at once: We witness some kind of an internal short circuit. In the case of a long lifetime, the electrons remain in the excited state long enough to be able to reach the surface of the crystal and to induce chemical processes.” Titania in the anatase form is particularly well suited for the chemical process purpose because it is provided with a special electronic structure that prevents “internal short circuits”.
Knowledge of this feature will allow the researchers to further optimize shape, size, and doping of anatase particles used inside photoreactors. The objective is to develop photoactive materials with higher efficiencies and longer lifetimes.
Professor Olaf Deutschmann, spokesman of the Helmholtz Research Training Group on ‘Energy-related Catalysis’ says, “The results obtained by Professor Wöll and his co-workers are of great importance regarding the generation of electrical and chemical energy from sunlight, and especially regarding the optimization of photoreactors.”
The results obtained by the researchers have been published in Physical Review Letters.
The prospects that catalysts will offer far cheaper fuel cells and that solar panels could generate hydrogen with a common and very low cost catalyst might offer household power, plus perhaps some vehicle charging. Solar going straight to chemical fuel, even hard to hold hydrogen as a store of the solar energy has a nice personal independence feel about it.
The European team is only at the first step, but the research isn’t about if it can be done, it’s about how it happens so that how to do it can be improved.
This week has seen good solar research progress for both the solar to hydrogen production step with a low cost active component for the catalyst and the low cost catalyst in the fuel cell for getting to electricity or heat or both. Solar to fuel and hydrogen to power has had a good week. They needed it.
Comments
3 Comments so far


See http://www.nickelpower.org to see how Dr. Rossi’s energy catalyzer will make energy obsolete, and to discover what the new world will look like.
[…] Titanium Oxide Solar Photocatalysis For Making Fuel | New Energy … Apr 28, 2011 … A European team has begun looking into the basics of titanium dioxide based photocatalysts for … […]
For your reference, titanium oxide has successfully been used for the solidified body for disposing of radioactive waste. If interested, please visit http://mid-autumn2008.blogspot.com/.