May
20
A New Fuel Cell Catalyst Breakthrough
May 20, 2009 | 3 Comments
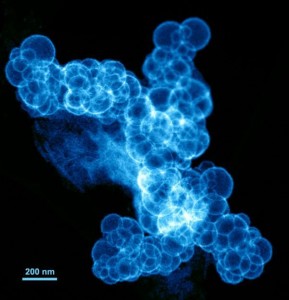
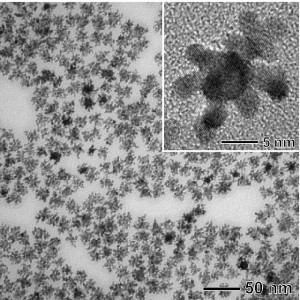
Younan Xia, Ph.D., the James M. McKelvey Professor of Biomedical Engineering at Washington University in St. Louis led a team of scientists at WUSTL and the Brookhaven National Laboratory in the development of a bimetallic fuel cell catalyst made of a palladium core that acts as a “seed” that supports growth of dendritic platinum branches like arms fixed to the palladium on the nanostructure. The size is only a nine-nanometer palladium core and seven-nanometer platinum arms. The technique makes a new bimetallic fuel cell catalyst that is efficient, robust and two-to-five times more effective than current commercially available catalysts.
The new and novel technique may in time enable a cost effective fuel cell technology that has been waiting for years for a breakthrough and should give a boost for much more efficient use of fuels worldwide.
The team synthesized the catalysts by sequentially reducing the precursor compounds to palladium and platinum in a liquid solution with L-ascorbic acid, common Vitamin C. The catalysts have a very high surface area, critical for catalyst efficiency and a number of applications besides fuel cells, plus the catalyst molecules are robust and stable.
The Department of Energy has estimated that widespread commercial success a fuel cells in the loading of the platinum, or the accumulation of debris, the catalyst’s loading should be reduced by four times in order to slash the operational costs of replacement. The WUSTL bimetallic catalyst is expected to substantially reduce the loading of platinum, be a more robust catalyst that won’t have to be replaced as often, and make better use of a very limited and very expensive supply of platinum.
The study was published and available in the online journal Science. Team participants include Byungkwon Lim, Pedro H. C. Camargo, Eun Chul Cho, Xianmao Lu, and Younan Xia at the Department of Biomedical Engineering, Washington University, Majiong Jiang at the Department of Chemistry, Washington University, and Jing Tao and Yimei Zhu at the Condensed Matter Physics & Materials Science Department, Brookhaven National Laboratory. A large diversified team indeed.
How good is it? Try this. Xia and the team tested how the catalysts performed for the oxygen reduction reaction process in a fuel cell. That determines how large a current will be generated in an electrochemical system as with the cathode of a fuel cell. They found that their bimetallic nanodendrite formed molecules at room temperature, were two-and-a-half times more effective per platinum mass for the process than state of the art commercial platinum catalysts and five times more active than other popular commercial catalysts. At 60 degrees C (the typical operation temperature of a fuel cell), the performance almost meets the targets set by the U.S. Department of Energy. This with the early discovery molecules. That’s mighty good.
Xia offers, “There are two ways to make a more effective catalyst. One is to control the size, making it smaller, which gives the catalyst a higher specific surface area on a mass basis. Another is to change the arrangement of atoms on the surface. We did both. You can have a square or hexagonal arrangement for the surface atoms. We chose the hexagonal lattice because people have found that it’s twice as good as the square one for the oxygen reduction reaction.” I suspect the team had some interesting conversations about their early choices or they have more investing in experimentation than made known publicly. However they got so far, it’s a huge payoff.
“We’re excited by the technique, specifically with the performance of the new catalyst,” Xia says. I dare say so. If this proves commercial, fuel cells have come a very long way in price and cost to compete in wide and common usage.
Xia says “growth seed start” has emerged recently as a good technique for precisely controlling the shape and composition of metallic nanostructures prepared in liquid solutions. It’s the only technique that allowed Xia and his team to come up with their unconventional shape.
Xia addresses another important property of the molecules, “When you have something this small, the atoms tend to aggregate (together) and that can reduce the surface area. A key reason our technique works is the ability to keep the platinum arms fixed. They don’t move around. This adds to their stability. We also make sure of the arrangement of atoms on each arm, so we increase the activity.” There’s another key, a stable catalyst molecule. It’s definitely breakthrough quality work.
The team is now exploring the possibility of adding other noble metals such as gold to the bimetallic catalysts, making them trimetallic. Gold has been shown to oxidize carbon monoxide, perhaps making for even more robust catalysts that can resist the poisoning by carbon monoxide — a reduction byproduct of some fuels like methanol or ethanol. If they can manage a breakthrough at that, the matter for liquid fuels to go the fuel cell route over internal combustion would be very compelling. Xia says, “Gold should make the catalysts more stable, durable and robust, giving yet another level of control.”
The outside major matter to consider is the incredible prices these metals demand. Palladium, platinum and gold are all very expensive. But perhaps the lessons from catalytic converters in autos, where precious metals have been greatly reduced on a per car basis and post service life recycling, will have a moderating effect. There is also the fact that the precious metals in an internal combustion drive wouldn’t be made so lessening some demand pressure.
The fuel cell offers a way to electric drive, recharging, storage and greatly improving the power and drive gear choices in transport vehicles. When I recheck the numbers claimed, the WUSTL team has essentially brought fuel cells to nearly practical engineering as consumers would expect in personal transport vehicles.
The breakthrough choice or perspective comes from this writer’s sense for a long time – that fuel cells have a very long way to go.
That gap has just been closed in a large way. Let’s hope for ease in getting to scale and more innovation and development. Fuel cell technology is still young, but getting to true viable marketability will sure push things along.
Comments
3 Comments so far


Hello, can you please post some more information on this topic? I would like to read more.
What a great resource!
thanks admin..good post