Sep
25
A Major Hydrogen Production Milestone
September 25, 2008 | 3 Comments
Whether one is dreaming of a hydrogen economy or looking for hydrogen to support carbon fuel production and carbon recycling the news out of the Idaho National Laboratory is the good stuff.
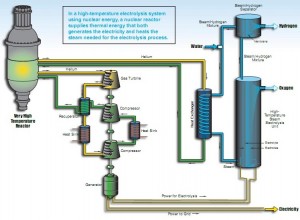
Reversing the technology of a hydrogen fuel cell where the hydrogen and oxygen combine to make electricity the INL group uses the electricity to drive the “fuel cell” so that it splits water into the hydrogen and oxygen. It’s a form of electrolysis, with very different electrodes and run at high temperature.
The system could be run from a high temperature nuclear reactor that would offer heat both to make steam for powering generation and heating the water prior to entering electrolysis. The milestone is the solid oxide fuel cell material has proven feasible and functions at a rate of 5.6 cubic meters per hour. As lab bench kits go, that is a lot of hydrogen if the pressure measurement isn’t too low.
The INL calls the experimental unit that set the milestone an Integrated Laboratory Scale plant (ILS). Operating at 800 degrees Celsius driven to full output later this year the yield should come in at 500 grams of hydrogen per hour. Stephen Herring, the INL nuclear physicist who heads up the High-Temperature Electrolysis project, says, “That doesn’t sound like much, but hydrogen is light stuff.” The lessons the team learns with the ILS will help them design a full-scale plant capable of producing two-and-a-half kilograms of hydrogen each second. Note that kilogram of hydrogen contains roughly the same amount of energy as a gallon of gasoline, and the world burns through about 120,000 gallons of oil a second.
To produce hydrogen the team uses fuel cell-like materials. In each solid oxide cell, a voltage pulls oxygen ions through a ceramic electrolyte, effectively separating the steam into hydrogen and oxygen. The team is working with Ceramatec, Inc. in Salt Lake City to produce the cells.
Experimentation began four years ago with a single, small button cell about an inch in diameter. Since then, the team has changed the geometry of the cells from single buttons to a series of stacks. Steam is pushed through every other layer of stacks and is split to produce hydrogen and oxygen. On the other side of the ceramic membrane, the oxygen ions that have migrated through the ceramic electrolyte are carried away using normal air. The geometric change from single cells to stacks was a crucial step. “We’ve gone up a factor of 15,000 in hydrogen production from the little button cells we had to what we have now,” Herring says, “but we have another factor of 15,000 still to go.”
The ILS experiment will incorporate 720 cells that will fit in a hotbox the size of a steamer trunk. When running at full power, the ILS will consume 15 kW of energy to power the electrolysis cells that will produce its hourly 500 grams of hydrogen. The team is working on ways to reduce that energy by making the electrolysis cells more efficient. They also plan to add heat exchangers to transfer heat from the end of the ILS to heat up water at the beginning. Creating that cyclical process will reduce the power needed to make steam, enabling the system to use 20 percent less electricity than it does now.
As one can imagine in a hot environment with a lot of free oxygen, oxidizing corrosion is a problem resulting in reduced efficiency. The team is up to 2000 hours of continuous operations or three months. For commercial use the cells have to be developed to last two years. At the next dismantle the used cells will go to the Argonne National Laboratory, the University of Nevada, and Ceramatec in Salt Lake for examination and added input in further development.
Regular readers will know that a foundation problem for endless supplies of fuels is free hydrogen. Hooked to carbon through a variety of ways free hydrogen solves most all petroleum, fuel and energy problems when the source energy to free the hydrogen is available at low enough cost. Here INL has come a long way, direct from nuclear to freed hydrogen and on to carbon based fuels as we’ve seen to date.
As we covered a couple of days ago in the Congress’ miss-named energy bill you might note that there isn’t any Federal assistance to free hydrogen, driving down the costs of fission nuclear power or research money for fusion nuclear power.
Which leaves the well informed wondering, can these levels of innovation and intellectual prowess from the likes of the INL scientists pay off for everyone else? The next key is going to be from the fusion camp or an intense effort to drive down fission plant costs.
With carbon readily reactive, available essentially everywhere, free hydrogen opens the door to energy security or independence if you prefer. Made into anything from methane to power fuel cells, heat homes or fuel automobiles on up to diesel and jet fuel, free hydrogen in cheap, big amounts should be a primary national goal.
Comments
3 Comments so far




[…] Go to the author’s original blog: A Major Hydrogen Production Milestone […]
[…] Made into anything from methane to power fuel cells, heat homes or fuel automobiles on up to diesel and jet fuel, free hydrogen in cheap, big amounts should be a primary national goal. ShareThis. Read More […]
The Idaho National Laboratory has a lot of other environmentally friendly projects. The Web site is http://www.inl.gov but there is also a channel at YouTube.
http://www.youtube.com/user/IdahoNationalLab
I would suggest the “Motion to Energy M2E” or “Harvesting the Sun’s Energy” videos.