Jan
28
A New Catalyst For Ethanol Fuel Cells
January 28, 2009 | 3 Comments
Scientists at the U.S. Department of Energy’s Brookhaven National Laboratory, collaborating with researchers from the University of Delaware and Yeshiva University, have developed a new catalyst that could make ethanol-powered fuel cells feasible. The highly efficient catalyst performs two crucial, and previously unreachable steps needed to oxidize ethanol and produce clean energy in fuel cell reactions. Until now scientists haven’t found a catalyst capable of breaking the bonds between ethanol’s carbon atoms. The new catalyst efficiently oxidizes the carbon atoms into carbon dioxide and does it at moderate temperatures, about room temperature in fact.
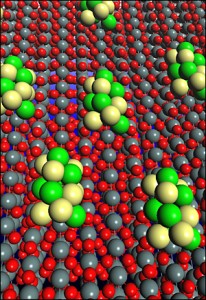
The new catalyst is made of platinum and rhodium atoms on carbon-supported tin dioxide nanoparticles. The structural and electronic properties of the new electrocatalyst were determined using powerful x-ray absorption techniques at Brookhaven’s National Synchrotron Light Source, combined with data from transmission electron microscopy analyses at Brookhaven’s Center for Functional Nanomaterials. Based on these studies and calculations, the researchers predicted that the high activity of their ternary catalyst resulted from the synergy between all three constituents – platinum, rhodium, and tin dioxide – knowledge that could be applied to other alternative energy applications.
Brookhaven chemist Radoslav Adzic says, “Ethanol is one of the most ideal reactants for fuel cells. It’s easy to produce, renewable, nontoxic, relatively easy to transport, and it has a high energy density. In addition, with some alterations, we could reuse the infrastructure that’s currently in place to store and distribute gasoline.” Volume and scale wouldn’t be new market startup issues.
Ethanol fuel cells convert hydrogen and oxygen into water and, during the process, produce electricity from the motion of the hydrogen electrons from one side of a cell to the other. The waste material is water and CO2, which are both sourced from and part of a natural biomass recycling system. Ethanol as a fuel cell fuel answers most all the near term hydrogen problems, and is very hydrogen rich. Energy density a familiar term to many, is a little different when looking at fuel cells. In these systems the density and availability of the hydrogen is paramount, the carbon is simply a hydrogen stabilizer and storage medium.
Adzic goes on saying, “The ability to split the carbon-carbon bond and generate CO2 at room temperature is a completely new feature of catalysis. There are no other catalysts that can achieve this at practical potentials. These findings can open new possibilities of research not only for electrocatlysts and fuel cells but also for many other catalytic processes.”
Next, the researchers will test the new catalyst in a real fuel cell in order to observe its unique characteristics first hand. But problems remain; the obvious is the use of rare and expensive platinum. The other is that methanol fueled cells have no need to split the carbon as it is a single carbon atom vs. the two carbon atoms in ethanol. It looks like there could be some competition in the future.
The paper is published online in the January 25, 2009 edition of Nature Materials and funded by the Office of Basic Energy Sciences within DOE’s Office of Science. It’s another step closer to electrical power on demand from high efficiency fuel cells with a commonly available fuel.
Comments
3 Comments so far


[…] Here is the original: A New Catalyst For Ethanol Fuel Cells | New bEnergy/b and Fuel […]
[…] the progress in fuel cells that split out the carbon in ethanol suggesting models to come, a story about Nissan that they have a solid oxide fuel cell that may be […]
Real informative and good structure of articles, now that’s user pleasant (:.