May
30
Convert Natural Gas to Liquid With Bacteria
May 30, 2019 | 1 Comment
Northwestern University researchers have found the enzyme responsible for the methane-methanol conversion in methanotrophic bacteria. The bacteria catalyzes the reaction at a site that contains just one copper ion. This finding could lead to newly designed, human-made catalysts that can convert natural gas to readily usable methanol with the same effortless mechanism.
Known for their ability to remove methane from the environment and convert it into a usable fuel, methanotrophic bacteria have long fascinated researchers. But how, exactly, these bacteria naturally perform such a complex reaction has been a mystery.
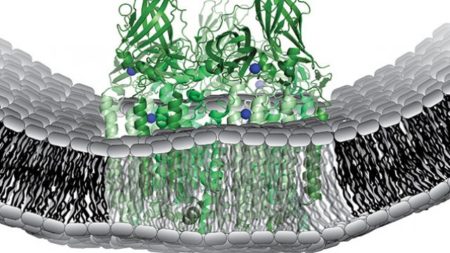
Schematic of the metabolic enzyme, particulate methane monooxygenase, which catalyzes the methane-to-methanol reaction. Image Credit: Northwestern University. Click image for the largest view.
Northwestern’s Amy C. Rosenzweig, co-senior author of the study said, “The identity and structure of the metal ions responsible for catalysis have remained elusive for decades. Our study provides a major leap forward in understanding how bacteria methane-to-methanol conversion.”
Brian M. Hoffman, co-senior author said, “By identifying the type of copper center involved, we have laid the foundation for determining how nature carries out one of its most challenging reactions.”
The study has been published in the journal Science.
Rosenzweig is the Weinberg Family Distinguished Professor of Life Sciences in Northwestern’s Weinberg College of Arts and Sciences. Hoffman is the Charles E. and Emma H. Morrison Professor of Chemistry at Weinberg.
By oxidizing methane and converting it to methanol, methanotrophic bacteria (or “methanotrophs”) can pack a one-two punch. Not only are they removing a harmful greenhouse gas from the environment, they are also generating a readily usable, sustainable fuel for automobiles, electricity and more.
Current industrial processes to catalyze a methane-to-methanol reaction require tremendous pressure and extreme temperatures, reaching higher than 1,300° Celsius. Methanotrophs, however, perform the reaction at room temperature and “for free.”
Matthew O. Ross, a graduate student co-advised by Rosenzweig and Hoffman and the paper’s first author explained, “While copper sites are known to catalyze methane-to-methanol conversion in human-made materials, methane-to-methanol catalysis at a monocopper site under ambient conditions is unprecedented. If we can develop a complete understanding of how they perform this conversion at such mild conditions, we can optimize our own catalysts.”
It would be a boon to stranded natural gas fields and turn flaring into product and profit as well as reduce methane’s heat holding effect in the atmosphere. If this scales well, the natural gas hydrate reserves around the world might become usable. This might be a very big thing and soon.
Comments
1 Comment so far


This is HUGE.
The single biggest problem with Natural Gas is storing and transporting it as a Gas. This discovery would give gas producers an entirely different option. Plus, making flaired gas into a valuable revenue stream would have drillers smiling all the way to the bank.