Feb
26
A Sodium Ion Battery Boost Found
February 26, 2019 | Leave a Comment
Nagoya Institute of Technology scientists have found the desirable component for sodium-ion batteries (SIB), which could contribute to boost SIB performance such as speed of charge. While lithium-ion batteries are currently popular, it is expected that a new storage chemistry will be found soon, because lithium is not only expensive but limited in supply. The findings show the possibility that SIB will be an attractive alternative for the lithium ion batteries.
The findings were published in Scientific Reports. The research effort was headed by Naoto Tanibata, Ph.D., an Assistant Professor at the Department of Advanced Ceramics at NITech.
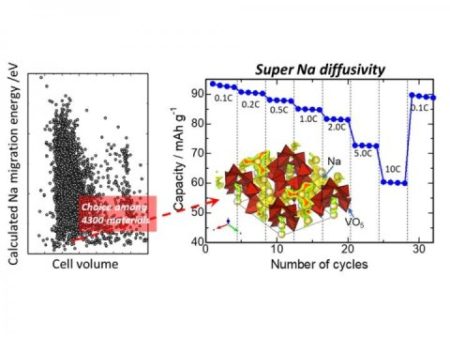
A high-throughput computation for Na migration energies is conducted for about 4,300 compounds in the inorganic crystal structure database, which the compound indeed exhibited excellent high-rate performance and cyclic durability; in detail, the compound exhibits stable 10C cycling, which corresponds to the rate of only six minutes for full charge/discharge, and ca. 94 percent capacity retention after 50 charge/discharge cycles at room temperature. These results are comparable with or outperform representative cathode materials for sodium ion batteries. Image Credit: Nagoya Institute of Technology. Click image for the largest view.
Popular lithium-ion batteries have several benefits – they are rechargeable and have a wide application spectrum. They are used in devices such as laptops and cell phones as well as in hybrid and fully electric cars. The electric vehicle – being a vital technology for fighting pollution in urban areas as well as ushering in clean and sustainable transport is an important player in the efforts to solving sustainable energy and environment conditions. One downside to lithium is the fact that it is a limited resource. Not only is it expensive, but its annual output is (technically) limited (due to the drying process). Given increased demand for battery-powered devices and particularly electric cars, the need to find an alternative to lithium – one that is both cheap as well as abundant – is becoming more urgent.
Sodium-ion batteries are an attractive alternative to lithium-based ion batteries for several reasons. Sodium is not a limited resource – it is abundant in the earth’s crust as well as in seawater. Also, sodium-based components have a possibility to yield much faster charging time given the appropriate crystal structure design. However, sodium cannot be simply swapped with lithium used in the current battery materials, as it is a larger ion size and a slightly different chemistry. Therefore, researchers are requested to find the best material for a sodium ion battery among vast number of candidates by a trial-and-error approach.
The scientists at NITech have found a rational and efficient way around the trial-and-error issue. After extracting about 4300 compounds from the crystal structure database and following a high-throughput computation of said compounds, one of them yielded favorable results and was therefore a promising candidate as a sodium-ion battery component. The researchers identified that Na2V3O7 demonstrates desirable electrochemical performance as well as stable crystal and electronic structures. This compound shows fast charging performance, as it can be stably charged within 6 minutes. Additionally, the researchers demonstrated that the compound leads to long battery life as well as the short charging time.
Dr. Tanibata said, “Our aim was to tackle the biggest hurdle that large-scale batteries face in applications such as electric cars that face long charge durations. We approached the issue via a search that would yield materials efficient enough to increase a battery’s rate performance.”
Despite the favorable characteristics and overall desired impact on sodium-ion batteries, the researchers found that Na2V3O7 underwent deterioration in the final charging stages, which limits the practical storage capacity to half of the theoretical one. As such, in their future experiments, the researchers aim to focus on improving the performance of this material so that it can remain stable throughout the entire duration of the charging stage.
“Our ultimate goal is to establish a method that will enable us to efficiently design battery materials via a combination of computational and experimental methods,” Dr. Tanibata added.
Its a remarkable achievement in speeding up a solution to a problem that has several years of effort already done without a viable new chemistry working at commercial scale.
One could say this team is almost there, but maybe they are fully halfway. But it does suggest that the improvement are going to come – and at a much faster pace.

