Mar
5
New Non Metallic Fuel Cell Catalyst
March 5, 2015 | Leave a Comment
Case Western Reserve University (CWRU) researchers have made an inexpensive metal-free catalyst. The new catalyst performs as well as costly metal catalysts at speeding the oxygen reduction reaction in an acidic fuel cell, and is more durable.
The study was recently published online in the journal Science Advances.
The carbon based catalyst is made of sheets of nitrogen-doped graphene that provides great surface area, carbon nanotubes that enhance conductivity, and carbon black particles that separate the layers allowing the electrolyte and oxygen to flow freely, which greatly increased performance and efficiency.
The CWRU group pursued a non-metal catalyst to perform in acid because the standard bearer among fuel cells, the PEM cell, uses an acidic electrolyte. PEM stands for both proton exchange membrane and polymer electrolyte membrane, which are interchangeable names for this type of cell that are the most common fuel cell type being tested and used in cars and stationary power plants.
For nearly half a century scientists have been trying to replace precious metal catalysts in fuel cells. Now the CWRU team has shown that an inexpensive metal-free catalyst performs as well as costly metal catalysts at speeding the oxygen reduction reaction in an acidic fuel cell. The carbon-based catalyst also corrodes less than metal-based materials and has proved more durable.
Liming Dai, the Kent Hale Smith Professor of macromolecular science and engineering at Case Western Reserve and senior author of the research said, “This definitely should move the field forward. It’s a major breakthrough for commercialization.”
Dai worked with lead investigator Jianglan Shui, who was a CWRU postdoctoral researcher and is now a materials science and engineering professor at Beihang University, Beijing; PhD student Min Wang, who did some of the testing; and postdoctoral researcher Fen Du, who made the materials. The effort builds on the Dai lab’s earlier work developing carbon-based catalysts that significantly outperformed platinum in an alkaline fuel cell.
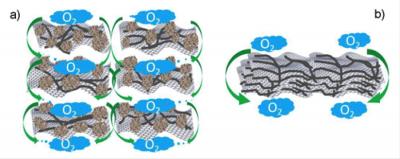
The key to the new catalyst is its rationally designed porous structure, Dai said. The researchers mixed sheets of nitrogen-doped graphene, a single-atom thick, with carbon nanotubes and carbon black particles in a solution, then freeze-dried them into composite sheets and hardened them.
Graphene provides enormous surface area to speed chemical reactions, nanotubes enhance conductivity, and carbon black separates the graphene sheets for free flow of the electrolyte and oxygen, which greatly increased performance and efficiency. The researchers found that those advantages were lost when they allowed composite sheets to arrange themselves in tight stacks with little room between layers.
Testing showed the porous catalyst performs better and is more durable than the state-of-the-art nonprecious iron-based catalyst. Dai’s lab continues to fine-tune the materials and structure as well as investigate the use of non-metal catalysts in more areas of clean energy.
We’ve seen a lot of catalyst breakthroughs over the years. What happens to them is sheer speculation. Platinum is still dominating at this writing. Your humble writer wishes the best for the team and congratulates them on their successful work. The hard part is yet to come.