Jan
15
Solar Powered Hydrogen Production Gets a Great New Idea
January 15, 2014 | 2 Comments
Meyer, Arey Distinguished Professor of Chemistry at UNC’s College of Arts and Sciences said, “So called ‘solar fuels’ like hydrogen offer a solution for how to store energy for nighttime use by taking a cue from natural photosynthesis. Our new findings may provide a last major piece of a puzzle for a new way to store the sun’s energy – it could be a tipping point for a solar energy future.”
The UNC team is reportedly motivated because solar energy has long been used as a clean alternative to fossil fuels such as coal and oil, but it could only be harnessed during the day when the sun’s rays were strongest. Their goal is said to be allowing us to power our devices long after the sun goes down.
The press release uses the suspicious idea that over one hour, the sun puts out enough energy to power every vehicle, factory and device on the planet for an entire year. It might be the earth receives that much energy. Thus solar panels can harness that energy to generate electricity during the day. But the problem with the sun is that it goes over the horizon at night – and with it the ability to power our homes and cars. If solar energy is going to have a shot at being a clean source for powering the planet, scientists have to figure out how to store it for nighttime use.
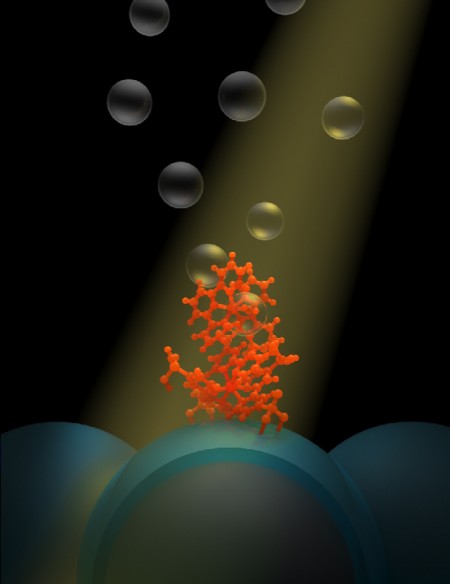
The new system designed by Meyer and colleagues at UNC and with Greg Parsons’ group at North Carolina State University does exactly that. It is known as a dye-sensitized photoelectrosynthesis cell, or DSPEC, and it generates hydrogen fuel by using the sun’s energy to split water into its component parts. After the split, hydrogen is sequestered and stored, while the byproduct, oxygen, is released into the air.
Meyer explains the problems, “But splitting water is extremely difficult to do, you need to take four electrons away from two water molecules, transfer them somewhere else, and make hydrogen, and, once you have done that, keep the hydrogen and oxygen separated. How to design molecules capable of doing that is a really big challenge that we’ve begun to overcome.”
Meyer has been investigating DSPECs for years at the Energy Frontier Research Center at UNC and in prior experience. His newest design has two basic components: a molecule and a nanoparticle. The molecule, called a chromophore-catalyst assembly, absorbs sunlight and then kick starts the catalyst to rip electrons away from water. The nanoparticle, to which thousands of chromophore-catalyst assemblies are tethered, is part of a film of nanoparticles that shuttles the electrons away to make the hydrogen fuel.
But even with the best of attempts in the earlier efforts, the system always crashed because either the chromophore-catalyst assembly kept breaking away from the nanoparticles or because the electrons couldn’t be shuttled away quickly enough to make hydrogen.
To solve the two problems, Meyer turned to the Parsons group to use a technique that coated the nanoparticle, atom by atom, with a thin layer of a material called titanium dioxide. By using ultra-thin layers, the researchers found that the nanoparticle could carry away electrons far more rapidly than before, with the freed electrons available to make hydrogen. They also figured out how to build a protective coating that keeps the chromophore-catalyst assembly tethered firmly to the nanoparticle, ensuring that the assembly stayed on the surface.
With electrons flowing freely through the nanoparticle and the tether stabilized, Meyer’s new system can use the sun’s energy for making hydrogen fuel while needing almost no external power to operate and releasing no greenhouse gases. What’s more, the infrastructure to install these sunlight-to-fuel converters is in sight based on existing technology.
“When you talk about powering a planet with energy stored in batteries, it’s just not practical,” said Meyer. “It turns out that the most energy dense way to store energy is in the chemical bonds of molecules. And that’s what we did – we found an answer through chemistry.”
A next target is to use the same approach to reduce carbon dioxide, a greenhouse gas, to a carbon-based fuel such as formate or methanol.
Apparently Meyer and Parsons are content with the progress. There doesn’t seem to be a working lab bench unit or a prototype. Just how efficient the idea might be isn’t discussed. But the team looks close to a working solution that might be waiting for someone else to take up the research.
Its looks like the chemical fundamentals are worked out and the idea needs more engineering. Perhaps one of you will follow up . . .
Comments
2 Comments so far


I’ve tossed the idea around to mimic nature in the photosynthesis process or to take carbon dioxide, water and energy to produce a hydrocarbon and oxygen.
Work at UNC sounds exciting.
Hydrogen is a poor repository of energy. Methane or methanol would be far better in many ways.