May
9
A New Process to Make Steel
May 9, 2013 | 2 Comments
If you’ve seen photos or videos of the huge red-hot cauldrons in which steel is made, fueled by large amounts of energy that smoke and burn away impurities wouldn’t be surprised to learn that steel making is one of the world’s leading industrial users of energy and produces a nasty mix of effluent gas and particulates.
Sadoway’s new process offers two side benefits: The resulting steel should be of higher purity, and eventually, once the process is scaled up, a lower operating cost.
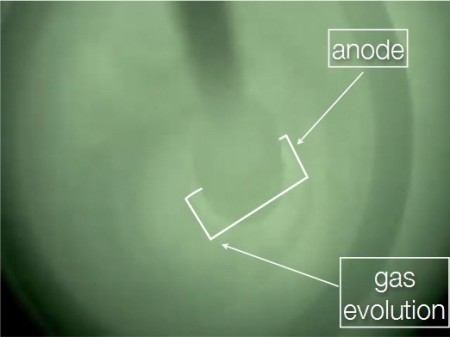
Molten Oxide Electrolysis in Action. Electrolysis cell viewed from the top at 1565° C, with the crucible wall represented by the dark circle and the anode location sketched in white. There is a video at the Nature link that follows. Click image for the largest view.
Worldwide steel production currently totals about 1.5 billion tons per year. The prevailing process makes steel from iron ore in the form of mostly iron oxide by intense heating with coal and blasts of air to uprate the temperature. The process forms carbon dioxide as a byproduct. Production of a ton of steel generates almost two tons of CO2 emissions, according to steel industry figures, accounting for as much as 5% of the world’s total greenhouse-gas emissions.
Additionally the process produces slag, ash and lots of particulate matter that is costly to remove and find uses for.
The industry has met little success in its search for carbon-free methods of manufacturing steel. The idea for the new method, Sadoway says, arose when he received a grant from NASA to look for ways of producing oxygen on the moon – a key step toward future lunar bases.
Sadoway’s method used an iridium anode, but since iridium is expensive and supplies are limited, that’s not a viable approach for bulk steel production on Earth. But after more research and input from Allanore, the MIT team identified an inexpensive metal alloy that can replace the iridium anode in molten oxide electrolysis.
It wasn’t an easy problem to solve, Sadoway explains, because a vat of molten iron oxide, which must be kept at about 1600 degrees Celsius, “is a really challenging environment. The melt is extremely aggressive. Oxygen is quick to attack the metal.”
Many researchers had tried to use ceramics, but these are brittle and can shatter easily. “I had always eschewed that approach,” Sadoway says.
Allanore takes up the explanation, “There are only two classes of materials that can sustain these high temperatures – metals or ceramics.” Only a few metals remain solid at these high temperatures, so “that narrows the number of candidates,” he says.
Allanore, who worked in the steel industry before joining MIT, says progress has been slow both because experiments are difficult at these high temperatures, and also because the relevant expertise tends to be scattered across disciplines. “Electrochemistry is a multidisciplinary problem, involving chemical, electrical and materials engineering,” he says.
The anode problem was solved using an alloy that naturally forms a thin film of metallic oxide on its surface: thick enough to prevent further attack by oxygen, but thin enough for electric current to flow freely through it. The answer turned out to be an alloy of chromium and iron – constituents that are “abundant and cheap,” Sadoway says.
The main benefit is the process produces no emissions other than pure oxygen and the process lends itself to smaller-scale factories: Conventional steel plants are only economical if they can produce millions of tons of steel per year, but this new process could be viable for production of a few hundred thousand tons per year, he says.
In addition to eliminating the emissions, the process yields metal of exceptional purity, Sadoway says. What’s more, it could also be adapted to carbon-free production of metals and alloys including nickel, titanium and ferromanganese, with similar advantages.
The third party observation chosen for the MIT press release, Ken Mills, a visiting professor of materials at Imperial College, London, says the approach outlined in the paper “seems very sound to me,” but he cautions that unless legislation requires the industry to account for its greenhouse-gas production, it’s unclear whether the new technique would be cost-competitive. Nevertheless, he says, it “should be followed up, as the authors suggest, with experiments using a more industrial configuration.”
Sadoway, Allanore and a former student have formed a company to develop the concept, which is still at the laboratory scale, to a commercially viable prototype electrolysis cell. They expect it could take about three years to design, build and test such a reactor.
It is also reported that the research was supported by the American Iron and Steel Institute and the U.S. Department of Energy. It’s good to see industry directly backing beneficial basic research.
The iron ore blast furnace has only been incrementally improved for decades since the idea burst forth over 100 years ago with a few standouts like the Swedish electric design that is known for very high quality steels.
The MIT team looks to have something quite a large evolutionary step ahead. With about 2000 years of humans working iron another big improvement is due. The mass involved in making iron ore to steel involves big numbers measured by millions of tons. Spreading the work out with no or very low pollution looks like a good step to get more competition, better quality and more innovative alloys.
The energy used will replace primarily coal with electric current. That opens the door to a drive to the lowest cost power generation. Let the battle begin driving down electricity and steel costs.
Comments
2 Comments so far


Environmentally minded countries have driven steel making to the 3rd world where pollution per ton is geometrically higher. This process could bring high quality, high tech steel back to the West. If it pans out, look for Germany to lead the way with this new process.
It’s going to be finish of mine day, but before end I am reading this enormous paragraph to improve my knowledge.