Aug
16
New Tool Doubles Production of Butanol and Cuts Costs
August 16, 2012 | Leave a Comment
A new discovery at the University of Illinois should make the alternative fuel butanol more attractive to the biofuel industry. Scientist Hao Feng has found a way to significantly reduce the cost of the energy involved and a way around a bottleneck that has frustrated producers in the past.
Feng explains, “The first challenge in butanol production is that at a certain concentration (level of butanol) the fuel being created becomes toxic to the organism used to make it (Clostridium pasteurianum and other strains), and that toxicity limits the amount of fuel that can be made in one batch. The second issue is the high energy cost of removing butanol from the fermentation broth at the high concentrations used by the industry. We have solved both problems.”
To achieve the study result, Feng’s team successfully tested the use of a non-ionic surfactant, or co-polymer, to create small structures that capture and hold the butanol molecules.
Feng adds, “This keeps the amount of butanol in the fermentation broth low so it doesn’t kill the organism and we can continue to produce it.”
The new process is called extractive fermentation and increases the amount of butanol produced during fermentation by 100% and peaks exceeding 200% have been recorded.
Using a range of non-ionic surfactants tests were ran to enhance the acetone–butanol (AB) production, and to extract and separate butanol from the fermentation broth. The best-optimized surfactant (L62) resulted in a butanol yield of 225% higher than the control. A considerable enrichment of butanol (6 times) was achieved in the surfactant-rich phase over the control. In addition, the downstream process volume or total liquids during extraction, was reduced by 4–6 times.
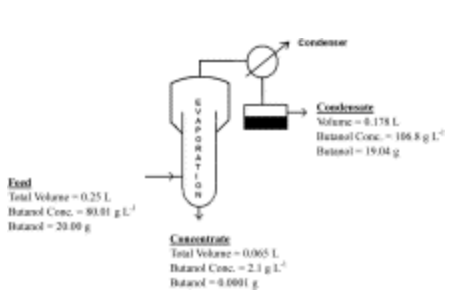
Butanol was separated from the surfactant-rich phase (obtained from model system) by evaporation between 120° and 130° C. The butanol was enriched in the condensate reaching a concentration of 106.8 g l−1, under which butanol automatically separated into two phases. The L62 was recovered by evaporation and reused for 3 recycles without affecting the partition coefficient, volume reduction, and butanol recovery in the surfactant-rich phase. The results demonstrated that the L62 not only significantly enhanced the butanol production but also functioned as a good extractant for separating butanol from the fermentation broth.
But that’s only the beginning. Feng’s group then makes use of one of the polymer’s properties – its sensitivity to temperature. When the fermentation process is finished, the scientists heat the solution until a cloud appears and two layers form.
Feng continues the explanation, “We use a process called cloud point separation. Two phases form, with the second facing the polymer-rich phase. When we remove the second phase, we can recover the butanol, achieving a three- to fourfold reduction in energy use there because we don’t have to remove as much water as in traditional fermentation.”
A bonus that the co-polymers have are they can be recycled and can be reused at least three times after butanol is extracted and recycling has little effect on phase separation behavior and butanol enrichment ability. After the first recovery, the volume of butanol recovered is slightly lower but is still at a high concentration.
According to Feng, alternative fuel manufacturers may want to take another look at butanol because it has a number of attractive qualities. Butanol has a 30 percent higher energy content than ethanol, lower vapor pressure, and is less volatile, less flammable, and mixes well with gasoline, he noted.
The study was funded by the Energy Biosciences Institute. The team members on Feng’s team are Pradip B. Dhamole, Zhilong Wang, Yuanqin Liu and Bin Wang.
It looks like this could be an excellent addition to the ethanol effort. While not mentioned or discussed, the vegetable material source to produce fuel isn’t shown as a factor. That implies indirectly that most all of the current feedstocks would work.
Butanol is a near drop in gasoline replacement. So far the production processes have simply been stopped by the concentration issue. Killing one’s fermentation bugs with the end product isn’t a path to success. If the Feng team can scale this work it shouldn’t be difficult to sign up some facilities and get the fuel into the market.
Lets hope so; butanol is a very good fuel.