Feb
20
New Electrolytes to Make an Ultra Flow Battery
February 20, 2012 | 3 Comments
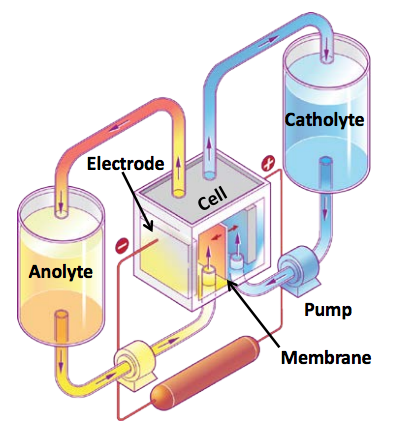
To start, a flow battery or accurately a Redox Flow Battery (RFB) is a type of rechargeable electrochemical system that relies on the redox states of various chemicals for the purpose of storing energy. The electroactive components, anolyte and catholyte, are liquids held in tanks. Charging and discharging of these batteries happens through a redox process in the cell. RFBs are attractive because in addition to the rapid charge/discharge ability is they can scale way up.
Process Graph of Metal Based Ionic Liquids used in a Flow Battery. Click image for the largest view.
To increase the energy stored in this type of system requires simply to only increase the size of the anolyte and catholyte tanks. Likewise, increasing the power output of the flow battery only necessitates the addition of more cells.
So far the problems have been charge cycle efficiencies, low energy densities, raw material costs, cross contamination of the anolyte and catholyte, plus corrosiveness and safety issues all have contributed to an unacceptably high cost per kWh.
The Sandia research published in Dalton Transactions, might lead to devices that can help economically and reliably incorporate large-scale intermittent renewable energy sources, like solar and wind, into the nation’s electric grid.
Anthony Medina, director of Sandia’s Energetic Components Realization program opens the discussion, “The U.S. and the world need significant breakthroughs in battery technology for renewable energy sources to replace today’s carbon-based energy systems. MetILs are a new, promising battery chemistry that might provide the next generation of stationary storage battery technology, replacing lead-acid and lithium-ion batteries and providing significantly higher energy storage density for these applications.” The electrical grid was designed for steady power sources, making fluctuating electricity from intermittent renewable energy difficult to accommodate. Battery based energy storage techniques can help even out the flow from fluctuating sources.
Sandia’s Travis Anderson is leading a team developing the next generation of flow batteries. In a flow battery a pump moves a solution of free-floating charged metal ions, dissolved in an electrolyte substance, with free-floating ions that conducts electricity, from an external tank through an electrochemical cell to convert the chemical energy into electricity. Flow batteries are rapidly charged and discharged by changing the charge state of the electrolyte. The electroactive material can be easily re-used many times. Anderson said flow batteries can sustain more than 14,000 cycles in the lab, equivalent to more than 20 years of energy storage. Sounds great.
But flow battery grid storage systems are roughly the size of a house and can cost more than equivalent lithium-ion batteries. So the Sandia group goal is to make flow batteries smaller and cheaper, while increasing the amount of energy stored for a given volume, or energy density. So far some flow batteries are out there, mostly using zinc bromine and vanadium redox, but they are moderately toxic and vanadium has big price fluctuations, plus they are vulnerable to the temperature. The Sandia group’s leaders hope to build a battery without water and solve these issues.
The Sandia team is multidisciplinary experts including electrochemist David Ingersoll, organic chemist Chad Staiger and chemical technologists Harry Pratt and Jonathan Leonard. What they’ve designed is a new family of electrochemically reversible, metal-based ionic liquids, or MetILs, which are based on inexpensive, non-toxic materials that are readily available within the U.S., such as iron, copper and manganese. They have a home run on new flow battery cathode materials.

Sandias Ionic Liquid Metal Salt Electrolyte Samples. MetILs are, from left to right: copper-based compound, cobalt-based compound, manganese-based compound, iron-based compound, nickel-based compound, and vanadium-based compound. Click the link above to the Sandia press release for more info and larger views.
Anderson takes up the explanation, “Instead of dissolving the salt into a solvent, our salt is a solvent. We’re able to get a much higher concentration of the active metal because we’re not limited by saturation. It’s actually in the formula. So we can cost-effectively triple our energy density, which drastically reduces the necessary size of the battery, just by the nature of the material.”
Here’s the payoff – The electrochemical efficiency, or ability to reverse charge, in MetILs is far greater than anything else published to date. The team has prepared nearly 200 combinations of cations, anions and ligands, and of those, five outperform the electrochemical efficiency of ferrocene, which has long been considered the efficiency gold standard.
The team has worked up the early solutions. Usually when mixing up positively and negatively charged molecules, the molecules will start clumping together, eventually causing the solution to turn gummy and clog up the battery membrane and electrode surfaces. The team solved that challenge by developing asymmetric cations, or positively charged ions, that resemble a soccer ball. In this analogy, the black pentagons represent negatively charged areas and the white hexagons represent positively charged regions. Such an arrangement lowers the melting point by preventing the ionic liquid constituents from bonding and becoming a solid, while the partial charge still allows electrons to flow freely through the cell to generate a current. It’s sort of “pre clumped” and works better for it – just don’t let it completely discharge.
Imre Gyuk, energy storage systems program manager at the Department of Energy’s Office of Electricity Delivery and Energy Reliability said, “The MetILs approach represents an ingenious, out-of-the-box solution to the cathode/electrolyte paradigm. Because it is based on readily available, inexpensive precursors, it may well lead to innovative, cost-effective storage systems with major impacts on the entire U.S. grid.”
Next up for the Sandia team is to find similar materials for the flow battery anodes. The team is encouraged by their progress so far.
Anderson said, “There are three things you’re juggling at the same time, and they aren’t always related: viscosity, electrical conductivity and the fundamental electrochemical efficiency. The excitement of having all three things go right at the same time, it’s like finding the treasure, but without the map. We’re creating that map, and we’re very excited by the possibilities.”
Lets see . . . low or non-toxic, 14,000 cycles, temperature tolerance, highest density so far, capacity by tank size . . . is anyone else thinking miniaturization?
Comments
3 Comments so far


Mr. Westenhaus, To avoid fallacious conclusions while looking for concrete progress, can you ask your fellow blogger with the bag over his head, (administrator theeestory) why eestor has not send any test sample to Sandia labs or Air Force Research Laboratory for testing? SL and AFRL have been waiting over 8 years for test samples from eestor.
fery good tenkyu
Fantastic.
Where I can buy this batteries for my electric scooter?