Jan
30
A Reduced Energy Way To Distill Alcohol
January 30, 2012 | 12 Comments
University of Manchester (UM) academics won the Nobel Prize in Physics in 2010 demonstrating graphene’s remarkable properties. Now Professor Sir Andre Geim’s team in a report published in Science shows that graphene-based membranes are impermeable to all gases and liquids as vacuum-tight. Except –
Water evaporates through graphene sheets prepared by the team as quickly as if the membranes were not there at all. The UM researchers have found that it is superpermeable with respect to water. That opens the possibility to produce alcohol without the energy input for heating the water and alcohol mix to drive the alcohol out in the separation. That would be a substantial energy input savings for ethanol production.
Graphene is one of the wonders of the science world, it’s the thinnest known material in the universe and the strongest ever measured. It conducts electricity and heat better than any other material. It is the stiffest one too and, at the same time, it is the most ductile.
The UN team studied membranes from a chemical derivative of graphene called graphene oxide. Graphene oxide is the same graphene sheet but it is randomly covered with other molecules such as hydroxyl groups OH-. The graphene oxide sheets are stacked on top of each other and form a laminate.
The researchers prepared such laminates that were hundreds of times thinner than a human hair but remained strong, flexible and are easy to handle. Then when a metal container was sealed with such a film, even the most sensitive equipment was unable to detect air or any other gas, including helium, to leak through.
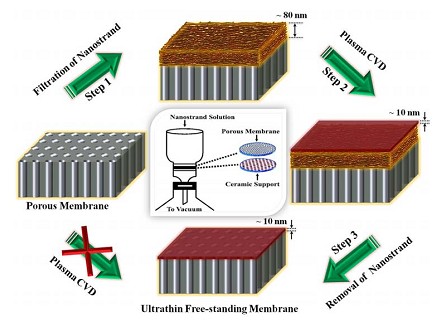
Graphene Sheet Water Filter Prep. Click image for more info, or see the Science link above for complete details.
When the researchers tried the same with ordinary water, they found with complete surprise that it evaporates without noticing the graphene seal. The water molecules diffused through the graphene-oxide membranes with such a great speed that the evaporation rate was the same independently whether the container was sealed or completely open.
Experiment leader Dr. Rahul Nair explains this way, “Graphene oxide sheets arrange in such a way that between them there is room for exactly one layer of water molecules. They arrange themselves in one-molecule thick sheets of ice, which slide along the graphene surface with practically no friction. If another atom or molecule tries the same trick, it finds that graphene capillaries either shrink in low humidity or get clogged with water molecules.”
Professor Geim follows up with, “Helium gas is hard to stop. It slowly leaks even through a millimeter-thick window glass but our ultra-thin films completely block it. At the same time, water evaporates through them unimpeded. Materials cannot behave any stranger. You cannot help wondering what else graphene has in store for us.”
Dr. Irina Grigorieva who also participated in the research points out the process advantage, “This unique property can be used in situations where one needs to remove water from a mixture or a container, while keeping in all the other ingredients.” The idea has impressive dewatering potential.
Dr. Nair throws in the ‘proof’ positive with, “Just for a laugh, we sealed a bottle of vodka with our membranes and found that the distilled solution became stronger and stronger with time. Neither of us drinks vodka but it was great fun to do the experiment.”
The vodka experiment made it to the research paper. Yet the team members are not offering visions of use in distilleries in particular, or offer any immediate ideas for applications. But we can just be certain serious attention is being given to the paper by ethanol researchers.
The basic research the UM team is doing does prompt a comment, albeit quite humble from Professor Geim, “The properties are so unusual that it is hard to imagine that they cannot find some use in the design of filtration, separation or barrier membranes and for selective removal of water.”
The graphene filters are not on the market or anticipated any time soon. The production cost of graphene for such a use isn’t even suggested or known but the research pathway to find out is now here. But the lifetime of this the new “filter” if such a term will do for now, looks to be very long indeed. And cutting the cost of heating the whole of the water and alcohol mix to get the water out is mostly removed, an awful lot of alcohol production that isn’t economic now, would be.
It’s very significant good news for the alterative renewable fuel folks. There’s lots more here than just ethanol potential, getting the water out cheaply is going to have lots of applications.
Comments
12 Comments so far



It sounds like these graphene membranes have potential in desalination and the distillation of brackish water as well.
My first thought exactly. Can these membranes be used as gravity sieves to desalinate seawater.
Sounds like the Reverse Osmosis process, as we now know it, could soon be history in the purification of water for potable water usages?
In the industrial chemistry world it’s called “separations”, and there’s lots of them. How about taking dilute solutions and making them concentrated for recycling. I can tell you, there’s many a waste stream that would instantly go marketwise if you could do cheap separations. Not to mention the throways that could be concentrated and snare the goodie, thus may find nice increases in yield. There’s money in it.
Wot’s the cost and stability, precious?
JP Straley
It will also revolutionise the moonshine industry in Ireland. Poitin “distillation” without a still or a heat source? Untraceable! That will drive the excise officers bananas. I wonder what graphene-extracted whiskey tastes like.
We’ll soon know, I imagine.
Craig,
I suspect that graphene-extracted whiskey taskes like grain alcohol (or potato alcohol) until you barrel it. Of course, you would still have to seperate the liquid from the mash. I guess you could squeese it out? Then remove the water with a graphene cover.
Matt
Scotch whisky distillers claim that even something as peripheral as the shape of the head of the still, and all sorts of other things that happen before casking, have radical effects on the final product.
They don’t seem to think their “nectar” is merely raw hooch flavoured by traces of left-over port, or something of the kind, imbued into the staves of a second-hand barrel.
The replacement of stills by graphene filtration would therefore make no small difference – unless the distillers are talking balderdash, that is.
I will be happy to sample all the hooch–excuse me, resultant product–from graphene separation.
Quick question: since the water molecule passes through the membrane after evaporation, isn’t it so that the energy required for the evaporation has already been consumed from the soroundings, meaning that in order to make this operational, heating of the water-ethanol mixture is necesary?
I realize that having no risk of getting ethanol through the membrane is a huge advantage, but I am thinking that the mixture needs to be heated to vaporation anyway. The heat would probably be reusable. Also the heat that the ethanol vapor has absorbed is kept on the mixture-side, adding to the potential efficiency of the process..
Nice post.Thank you for taking the time to publish this information very useful!I’m still waiting for some interesting thoughts from your side in your next post thanks.
[…] that a similar non-combustion distilling process is possible through the use of a material called graphene, a material associated with a nobel […]
Jene werden jedoch nicht sehr bestreiten, daß jeder angesichts jener zahlreich aufgetretenen Grippefälle auch in Deutschland welche
Sch – Hugo Sekt grippe nicht mit die „leichte Schulter” nehmen soll und jedoch nicht sehr alles lediglich Panikmache ist. Über jenen integrierten Hugo Sekt Bildeditor werden Diese selbst völlig einfach eine Nachbearbeitung der eigenen Bilder vornehmen auch die nur so optimal an jenes Etikett anpassen. Auch bei ganz besonderen Anlässen geht es sehr einfach nicht sehr ohne Hugo Sekt.