Dec
9
The Path From CO2 to Fuel Improves
December 9, 2010 | 5 Comments
After yesterday’s look at new field of chemistry using palladium to reform CO2 back to fuel University of Illinois’ scientists Dr. Paul Kenis and graduate student Devin Whipple published the accomplishments to date in the field of the electrochemical reduction of CO2 in the ACS Journal of Physical Chemistry Letters. As in yesterday’s post, the idea is to produce a variety of organic compounds for use as feedstocks for chemical synthesis or conversion into hydrocarbon fuels.
In this paper the authors point out substantial advances in catalysts, electrolytes, and reactor designs are needed to enable CO2 utilization via electrochemical conversion.
Kenis and Whipple note the electrochemical reduction of CO2 is one of the possible pathways under investigation for the conversion of CO2 to useful chemicals and fuels; others are photochemical or biochemical pathways – all areas in which significant progress is being made.
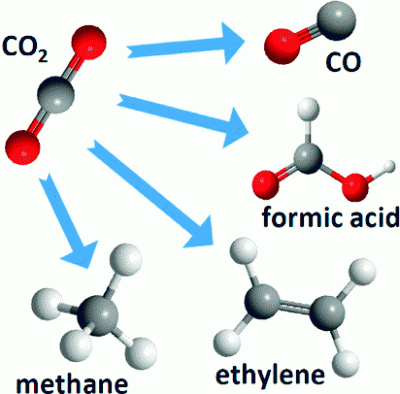
Electrochemical reduction is particularly attractive because direct heterogeneous electrochemical reduction of CO2 can produce several products, including formic acid, carbon monoxide, methane, ethylene, and methanol. The following graphic shows this clearly.
The best part of the paper is the consideration of potential. Two key areas spark considerable interest, the leveling the output from intermittent electricity sources such as wind and solar and allowing the production of liquid fuels for the transportation sector with renewable electricity.
By comparison, the electrolysis of water, which is essentially a mature technology, also allows storage of electricity in chemical form (hydrogen). But hydrogen does not fit into the existing infrastructure as easily as liquid fuels do, has a lower volumetric energy density, and requires costly investment and operations of compression or liquefaction for storage.
The paper does a good job in explaining the really big problem. Energy is needed and to soothe the green environmentalist crowd the electricity used to convert the CO2 must be renewable, or at least from a carbon-neutral source such as nuclear; otherwise, more CO2 could be emitted in producing the electricity than would be reduced in the process.
Yet the electrochemical conversion of CO2 has great potential to help overcome several of the challenges facing the implementation of carbon-neutral energy sources because it provides a means of storing renewable electricity in a convenient, high-energy-density form. Particularly exciting is the ability to produce methane and ethylene directly on catalysts and the possibility of producing even longer hydrocarbons from syngas. The guys at Illinois might want to talk with the folks at Washington University.
To be successful electrochemical conversion of CO2 needs to meet two criteria: (i) high energy efficiency and (ii) high reaction rates. High energetic efficiency – achieved through a combination of high selectivity (Faradaic or current efficiency) and low overpotentials- -is a critical parameter because it specifies the recoverable energy contained in the product; i.e., defines the energy cost of producing the product.
All reasonable and sensible.
Yet somehow conclusions are a significant part of the paper, which leads other observers to wonder – what is the point? This field is simply quite early in the research phase – a place that defies conclusions.
The highlight remains a valuable point – using renewable energy to reform CO2 is a great idea worth some serious interest especially if the product is simple methane or ethane for electricity production. That idea tied to a CO2 source could double, triple or make some of the carbon endlessly recyclable. The idea potential is amazing as seen in the comments for yesterdays post.
That portion makes the Kenis and Whipple paper worth a read. The rest – its premature judgments – already obsolete on the release date. Things are moving too fast.
Still, its getting clearer that somehow renewables are going to find a home where their value can pay off. Recycling CO2 might be a worthy answer over time.
Comments
5 Comments so far


Very energy intensive. Using nuclear power makes sense when coupled to a concentrated CO2 source such as a coal plant, cement plant etc. Wind and solar do not make economic sense on their own merits, much less for a second order application such as this, with necessary inefficiencies.
A previous commenter suggested a nuclear aircraft carrier at sea making its own jet fuel. But where would it get its CO2? The atmosphere? Only 0.04% CO2 there.
If you want jet fuel depots in the middle of the ocean, build seasteads that farm seaweed and convert the weed to chemicals, plastics, feeds, and fuel. Your seastead should be nuclear powered, with a SMR.
Actually, you could extract the CO2 from seawater, where concentration is a lot higher than in the atmosphere, and therefore should be easier.
Mind you, it will probably still be impractical and use obscene amounts of energy, but if you are going to use, say, nuclear power plants to extract hydrogen from water (via electrolysis or, even better, sulfur-iodine cycle) you will have to process huge volumes of water anyway. You might as well go the extra mile and extract your CO2 from the same place.
What can you say about it? I am not sure how a particular kind of energy would be useful for electricity, transportation, and other purposes. I personally need to know that futher.
But first, it is important to investigate the broadly philosopical & ideological phenomenon of what is known as “growth skepticism” mainly because of the increasing pessimism about the idea of economic growth and accompanying social progress.
Someone should have dealt with that now. Otherwise, they might end up with delusions that raise eyebrows of ordinary people around the globe, including me.
So please be warned about that. Thank you very much.
[…] This post was mentioned on Twitter by ulka melisa, Richard Pescod J.. Richard Pescod J. said: The Path From CO2 to Fuel Improves http://t.co/4NeVd7w vía @AddThis […]
Pete is correct. CO2 is often 200 times more prevalent in sea water than the atmosphere.
Ships already make freshwater at sea from sea water. With the right catalysts they could create a syngas that can be converted via Fischer Tropsch into fuel.
The key will be the catalysts.