Aug
27
Oxygen Splitting Breakthrough for Getting Free Hydrogen
August 27, 2010 | 10 Comments
That headline is accurate if a little bewildering. Splitting water to get hydrogen isn’t hard to do, it can be quite simple, and a little lab experiment on the table will do. But doing it the simple way gets you the combined hydrogen and oxygen gases in one mixture called Oxyhydrogen or Brown’s gas – a highly, very highly ignitable mixture of hydrogen and oxygen in the perfect mixture to recombine. That’s great if you immediately use it, but storing the gas is quite an engineering feat for the safety needed to avoid a very fast ignition and burn. Confined Oxyhydrogen gas isn’t something you want nearby if there’s any spark potential.
That makes the water splitting process much more desirable if the hydrogen and oxygen have their own electrodes and the freed gases come off separately. One can store hydrogen in a near pure state that must leak to get to oxygen for ignition and can only ignite with a proper mixture. That’s much, much safer.
MITs Daniel Nocera and his associates have found yet another formulation, based on inexpensive and widely available materials that can efficiently catalyze the splitting of water molecules using electricity in an electrolyzer. This form of electrolyzer uses two different electrodes, one of which releases the oxygen atoms and the other the hydrogen atoms. They described the advance at the 240th National Meeting of the American Chemical Society, being held in Boston this week.
Nocera’s report focused on the electrolyzer catalysts – materials that jumpstart chemical reactions like the ones that break water up into hydrogen and oxygen. Good catalysts already are available for the part of the electrolyzer that produces hydrogen. What are missing were inexpensive, long-lasting catalysts for the production of oxygen. Nocera’s new catalyst fills that gap and boosts oxygen production by 200-fold. It eliminates the need for expensive platinum catalysts and potentially toxic chemicals used in making them.
The new catalyst has already been licensed to newly formed Sun Catalytix, which envisions developing safe, super-efficient versions of the electrolyzer, suitable for homes and small businesses, within two years.
Nocera, along with postdoctoral researcher Mircea Dincă and graduate student Yogesh Surendranath, report the discovery is nickel borate, made from materials that are even more abundant and inexpensive than an earlier find. In 2008, Nocera reported the discovery of a durable and low-cost material for the oxygen-producing electrode based on the element cobalt.
Nocera is also pointing out a significant observation of his findings, that the original cobalt compound was not a unique, anomalous material, and suggests that there may be a whole family of such compounds that researchers can study in search of one that has the best combination of characteristics to provide a widespread, long-term energy-storage technology.
The research is still at the early stage. “This is a door opener,” Nocera says. “Now, we know what works in terms of chemistry. One of the important next things will be to continue to tune the system, to make it go faster and better. This puts us on a fast technological path.” While the two compounds discovered so far work well, he says, he is convinced that as they carry out further research even better compounds will come to light. “I don’t think we’ve found the silver bullet yet,” he says.
If you’re interested in getting in the hydrogen game Nocera is the guy to catch up with.
In the course of their research Nocera and his team have increased the rate of production from these catalysts a hundredfold from the level they initially reported on cobalt two years ago.
There are concentrated alkali based commercial scale electrolyzers of good efficiency now, but that kind of thing isn’t going work at small scale in residential, commercial, solar driven or remote, off grid kinds of locations. The alkali units need professional continuous oversight.
Nocera’s idea is more rational for the individual, “Our goal is to make each home its own power station,” says Nocera. “We’re working toward development of ‘personalized’ energy units that can be manufactured, distributed and installed inexpensively. There certainly are major obstacles to be overcome – existing fuel cells and solar cells must be improved, for instance.”
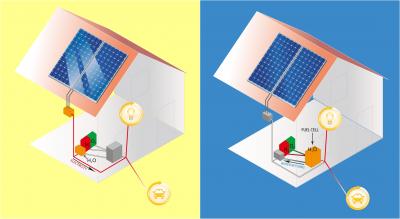
Such a system would consist of rooftop solar energy panels to produce electricity for heating, cooking, lighting, and to charge the batteries on the homeowners’ electric cars. Surplus electricity would go to the electrolyzer, to break down ordinary water into its two components, hydrogen and oxygen. Both would be stored in tanks. In the dark of night, when the solar panels cease production, the system would shift gears, feeding the stored hydrogen and oxygen into a fuel cell that produces electricity (and clean pure drinking water as a byproduct). Such a system would produce clean electricity 24 hours a day, seven days a week – even when the sun isn’t shining.
The technological barriers are cracking away for Nocera’s idea. The matter in not so long a time will likely be the capital investment needed to buy the systems. Its going to have to be cheap, and the solar cell crowd is getting there steadily if not allowing for wind, hail and hurricane weather. The biggest breakthrough will need to be in the fuel cell. That’s the main problem.
But there is great potential here. Nocera’s research isn’t knocking the concentrated alkali systems performance over – yet. But if he can, then mass production would drive to lower costs even faster.
It’s another race that is getting interesting to watch.
Comments
10 Comments so far


Insanity! To suggest this as a residential installation says someone is a few cards short of a full deck!
I would like to know you that the solar-hydrogen power system is still experimental in homes, residential & office buildings, and commercial & industrial facilities, etc.
It is clear that various types would emerge soon – but would it be possible to look into the merits of the solar-hydrogen technology?
However, please do not waste time on fighting it over on how this kind of power system works. And PLEASE, do not ever give up on doing experiments into one of these power supply systems.
It is time to roll up your sleeves, and get going! Good luck!
Thanks for sharing.
Very descriptive post.
May be you have info about most effective water turbine design ?
Quote, ‘I would like to know you that the solar-hydrogen power system is still experimental in homes, residential & office buildings, and commercial & industrial facilities, etc’.
Where – these are still concepts and lab pilot programs. I doubt anyone has one functioning in their home.
Safety first, last and always!
To “russ”:
Please discuss well about the definition of solar-hydrogen energy technology for residential, commercial & industrial buildings, OK?
It remains as an experiment (meaning concepts and lab pilot programs).
As you said to me:”Safety first, last and always”. (I would like you to think carefully what you said about this energy technology on your own merits, too.)
So always do what you must do regarding experimental energy technologies. Thank you very much.
From JPK
I can imagine the hard work it must have been required to research for this post. All what I can say is just keep publishing such posts, we all love it. And just to bring something to your notice, I have seen some blog providing your blog as source for their information.
I just signed up to your blogs rss feed. Will you post more on this subject?
It’s really a nice and helpful piece of information. I’m glad that you shared this helpful info with us. Please keep us informed like this. Thanks for sharing.
What a great resource!
Esta noticia es muy importante y valiosa para quienes creemos en el hidrógeno como un vector energético viable en el futuro inmediato. Es muy importante reconocer el esfuerzo que la ciencia hace hoy para hacer que lo complejo parezca muy simple. Pero, es todavía más valioso cuando las mentes que proponen estas nuevas ideas, piensan de manera altruista y desean hacer colectivo y viable su hallazgo para todo el público. La electrólisis de bajo costo, mejorando los catalizadores, electrodos y sobretodo electrolitos de manera que se produzca más hidrógeno, más puro e invirtiendo mucho menos energía. Felicito a Daniel y a su equipo de trabajo.