Dec
7
A New Enzyme to Break Free Hydrogen
December 7, 2009 | 3 Comments
A team from the University of Oxford in the UK and CNRS in France has developed an optimized system that will produce hydrogen gas.
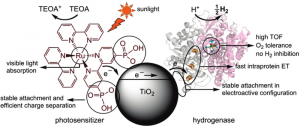
Powered by light’s energy and using an hydrogenase and photosensitizer co-attached to a TiO2 nanoparticle the lab sized apparatus production is 6 times higher than a process based in bimolecular (and probably) diffusion-controlled photo H2 production when replacing TiO2 by MV2.
The need to free hydrogen for use in fuel production is a major part of the journey to production of fuels in a current account instead of using the fossil fuel store. Hydrogen in a reactive state at low enough cost would make fuel production and use in a short-term cycle much more possible.
Oxford’s Dr. Fraser Armstrong and his colleagues selected the titaniaphilic enzyme Db [NiFeSe]-H (from the bacterium Desulfomicrobium baculatum) which is very stable even after prolonged exposure to air with a turnover frequency of 50 (mol H2) s-1 (mol enzyme)-1 upon visible light irradiation, when attached to a RuP-sensitized TiO2 at pH 7 and 25 °C (77 °F).
The paper says, “It thus provides a benchmark and reference on a per active site basis for future systems. Although this rate is lower than the maximum rate that hydrogenases can acquire, it is remarkably high considering the small driving force acting on the enzyme to produce H2 during irradiation. This direct-electron transfer controlled rate is 6 times higher than the one obtained by bimolecular (and probably) diffusion-controlled photo H2 production when replacing TiO2 by MV2+.”
Hydrogenases are natural enzymes used by some microbes in their energy metabolism that can reduce protons into hydrogen at active sites composed of iron- or nickel/iron complexes. The scientists note that the electrochemical reaction is reversible, and hydrogenases can produce or oxidize H2 with just a minimum overpotential—as does the expensive metal catalyst platinum.
The most interesting aspect of the research is the system shows high electrocatalytic stability not only under anaerobic conditions but also after prolonged exposure to air, thus making it sufficiently robust for benchtop applications. That observation of the experiment bodes well for increasing the scale. How far scale can go will be important as research continues.
How effective the system might be is hinted at in the paper’s abstract saysing, “The optimized system consisting of Db [NiFeSe]-H attached to Ru dye-sensitized TiO2, with triethanolamine as a sacrificial electron donor, produces H2 at a turnover frequency of approximately 50 (mol H2) s−1 (mol total hydrogenase)−1 at pH 7 and 25 °C, even under the typical solar irradiation of a northern European sky.
The deep research into bacteria is paying off. “The [NiFeSe]-hydrogenase from Desulfomicrobium baculatum (Db [NiFeSe]-H) is identified as a particularly proficient catalyst.”
What stimulates this writer’s interest is the operating conditions. 25C for people is just comfortable, not oustisde of the habitation range. The active fluid is only at a ph of 7, not a strong acid or base. Most curious is the experiments work under northern European skies, leaving out much of the solar intensity available from the experiment.
The question is if the research will get anywhere. Well informed and thoughtful people know the safest and elegant path, if research can close the technology gaps to endlessly recycled carbon fuels enriched with hydrogen, would be to use the planet and atmosphere to promote plant and animal life while using the system to cycle about the carbon for fuel use.
This European research effort gets us closer to seeing that paths are there to be found in the chemistry of life. Finding the most practical and economically viable will take time. But this news is exciting, knowing it can be done is a firm anchor for pressing on and discovering and learning more.
Photosynthesis might not need created in a single step; it may be all we need is the reactive part that cuts loose the hydrogen for a good start. What plants do in what seems to be a single process might be at industrial scale several process steps.
Comments
3 Comments so far



Valuable info. Lucky me I found your site by accident, I bookmarked it.
Have you ever considered about including a little bit
more than just your articles? I mean, what you say is fundamental and everything.
However imagine if you added some great pictures or video clips to give your posts more, “pop”!
Your content is excellent but with pics and clips, this site could definitely be one of the very best in its niche.
Good blog!
I love your blog.. very nice colors & theme.
Did you make this website yourself or did you hire someone to do it
for you? Plz answer back as I’m looking to construct my own blog and would like to find out where u got this from. appreciate it