Oct
31
A New Idea For Recovering Rare Earth Elements
October 31, 2013 | Leave a Comment
Scientists are reporting development of a new method to recycle rare earth elements from wastewater. Many of today’s technologies, from hybrid car batteries to flat-screen televisions, rely on rare earth elements (REEs) that are in short supply.
The process is described in a study in the journal ACS Applied Materials & Interfaces. The new process could help alleviate economic and environmental pressures facing the REE industry and favorably affect consumer goods prices over time.
Zhang Lin and colleagues point out that REEs, such as terbium – a silvery metal so soft it can be cut with a knife – behave in unique ways as super magnets, catalysts or superconductors. That makes them irreplaceable in many of today’s tech gadgets and machines.
Market watchers expect global demand to rise to at least 185,000 tons by 2015. Although some of these elements are actually plentiful, others are indeed in short supply. According to reports, terbium and dysprosium supplies may only last another 30 years. Attempts so far to recycle them from industrial wastewater are expensive or otherwise impractical. A major challenge is that the elements are typically very diluted in these waters.
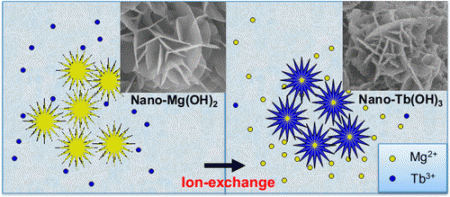
Lin’s team knew that a nanomaterial known as nano-magnesium hydroxide, or nano-Mg(OH)2, was effective at removing some metals and dyes from wastewater. So they set out to understand how the compound worked and whether it would efficiently remove diluted REEs, as well.
To test their idea, they produced inexpensive nano-Mg(OH)2 particles, whose shapes resemble flowers when viewed with a high-power microscope. They showed that the material captured more than 85 percent of the REEs that were diluted in wastewater in an initial experiment mimicking real-world conditions. In addition, a method was developed to further separate the immobilized REEs and the residual magnesium hydroxide by varying the solution pH.
“Recycling REEs from wastewater not only saves rare earth resources and protects the environment, but also brings considerable economic benefits,” the researchers state. “The pilot-scale experiment indicated that the self-supported flower-like nano-Mg(OH)2 had great potential to recycle REEs from industrial wastewater.”
The REE issue is of more importance to the world economy than most people realize. The elements are used in a wide array of devices and more and more devices have electronic components. REEs are used at ever increasing rates.
Everyone with a bit of sense should realize that just disposing to land fills items that contains recyclable materials is a way to increase future costs, not to mention the simple dirtying and trashing of our environment. For example, used lithium ion batteries are really not something one wants laying around disintegrating for important and very toxic reasons.
Recycling, while critical, isn’t going to increase the inventory of devices for sale or in use. For that more REE needs to be produced. The U.S. has a huge known inventory, but pressures of ‘not in my back yard’, government permitting and other barriers have production essentially stopped.
If you enjoy the latest technology, realize that a lot of life depends on existing technology that will need to be replaced and upgraded someday, or have an interest in these types of things and the power that makes them go, REEs are a vitally important thing to keep an eye on. It’s not a matter of strategic minerals, it’s continuing on with life as we know it.