Feb
14
A Catalyst Breakthrough With Organic Molecules
February 14, 2013 | 1 Comment
We usually think of catalysts as metals, often the rare and expensive platinum. So when chemists at Boston College announce they’ve designed a new class of organic molecule catalysts triggered by the charge of a single proton, one’s attention is seized. The team reports in the most recent edition of the journal Nature the new simple organic molecules offer a sustainable and highly efficient platform for chemical reactions that produce sets of molecules crucial to advances in chemistry, medicine and the life sciences.
As grand as the range of application is – we’re interested in fuels. The new catalysts form products consisting mainly of a single mirror-image isomer of a large assortment of amines and alcohols, which serve as building blocks for the preparation of molecules capable of becoming fuels, advancing new drug therapies relevant to human healthcare and the preparation of other life science products.
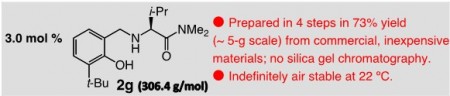
Organic Molecule Catalyst Molecule. For more info click the Nature link above. Click the image for a larger view.
The Boston College team’s work stands out because finding a reliable, truly general, efficient synthesis of single mirror-image isomers has proven elusive. Previous working methods suffer from a combination of extreme temperatures, long reaction times, limited scope, low selectivity, the need for rare or precious metals and highly toxic elements.
According to lead author Amir Hoveyda, the Joseph T. and Patricia Vanderslice Millennium Professor of Chemistry at Boston College the new catalysts are small organic molecules derived from the abundant and renewable amino acid valine and can be synthesized in four steps through the use of commercially available and inexpensive materials.
The catalyst, used in as little as one quarter of a percent, promotes reactions that are complete within two minutes to four hours typically at room temperature yielding the large assortment of amines and alcohols.
The electronic activation sparked by the proton and internal hydrogen bonds play a key role in every stage during catalysis of the carbon-carbon bond forming process, according to the researchers. This includes achieving high enantioselectivities – favoring one mirror-image isomer – as well as unprecedented rates of catalyst regeneration and product release.
The team reports small-molecule catalysts initiate reactions of readily available boron-containing reagents with easily accessible imines and carbonyls, producing amines and alcohols with a high degree of enantiomeric purity.
The catalyst’s ability to provide access to these prized enantiomerically enriched organic compounds in a manner that is not only efficient and selective but also economical and sustainable for the long term, will be of enormous value.
The co-authors include Boston College Professor of Chemistry Marc Snapper, Senior Research Associate Fredrik Haeffner, post-doctoral researchers Sebastian Torker and Tatiana Pilyugina, and graduate students Erika Vieira and Daniel Silverio.
A very proud Hoveyda said, “A reaction that can be initiated by a minute amount of a readily accessible and inexpensive catalyst to afford valuable organic molecules with high selectivity and which requires only renewable resources, as opposed to precious and rare elements, is extremely important to future advances in medicine and the life sciences.” Hoveyda is justified in the pride, but for commercial scale the team might want to look into the huge fuel potential to support the life science and medical uses.
Efficient, selective, cost-effective and sustainable protocols for preparation of organic molecules now can offer realistic access to significant quantities of a range of biologically active entities. Enantioselective synthesis, preparation of one mirror-image isomer, is crucial in this regard since most important entities in biology and medicine have the property of being handed as well.
The Boston College press release quotes Robert Lees, of the National Institutes of Health’s National Institute of General Medical Sciences, which partly funded the work as saying, “Chemical transformations that are highly selective as well as economical are very important for both discovery and commercial development of new therapeutic and diagnostic agents. The catalysts developed by Dr. Hoveyda represent an impressive advance because they can be used to inexpensively and predictably produce either isomer of a mirror image pair of molecules using mild reaction conditions.”
Hoveyda said the discovery will allow chemists to access many valuable organic molecules faster, cheaper and in a sustainable and economic fashion with minimal waste generation and without continuing to depend on diminishing reserves of precious metals.
“The new catalysts have all the key characteristics of a class of molecules that can serve as a blueprint for the invention of many additional important and useful reaction promoters in the future,” said Hoveyda.
Yup, Doc. That’s why you’re being covered at NewEnergyandFuels.com!
Comments
1 Comment so far


how to increase the oil produce rate