Feb
1
Hydrogen Production From Synthetic Catalysts
February 1, 2013 | Leave a Comment
Hydrogen has tremendous potential as an eco-friendly fuel, but its production requires lots of energy spoiling the ecological benefit in conventional production methods.
The team is led by Princeton’s David B. Jones Professor of Chemistry Annabella Selloni. The team’ innovation is using bacterial enzymes called di-iron hydrogenases that make hydrogen from water. Selloni’s team used computer models to figure out how to incorporate the bio processing of these enzymes into the design of practical synthetic catalysts that industry can use to produce hydrogen from water.
Whatever the process hydrogen production has the problem of the freed oxygen released during the separating reaction. In this latest paper, Selloni and co-authors present a solution by making changes to the catalyst to improve the stability of the structure in water. The researchers found that they had also created a catalyst that is tolerant to oxygen without sacrificing efficiency.
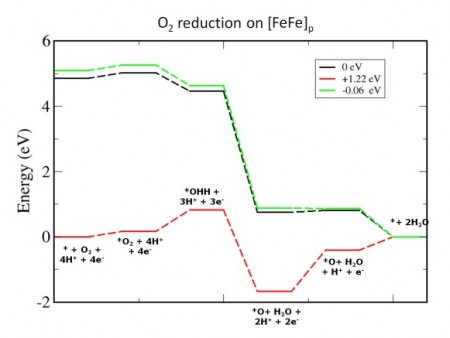
Hydrogen Evolution & Oxygen Reduction In Iron Based Electrochemical Catalyst. Click image for more info.
To frost the breakthrough their artificial catalyst could be made from abundant and cheap components, such as iron, indicating that the catalyst could be a cost-effective way of producing hydrogen.
So far though Selloni and her team have only conducted their research “in silico” – that is, using computer modeling.
The team expects to reach a goal of learning enough about how these catalysts work to someday create working catalysts that can make vast quantities of inexpensive hydrogen for use in vehicles and electricity production.
This is basic research that looks to have a very good future. Selloni’s team includes members Patrick H.L. Sit an associate research scholar in chemistry at Princeton; Roberto Car, Princeton’s Ralph W. *31 Dornte Professor in Chemistry, and Morrel H. Cohen, a Senior Chemist at Princeton and Member of the Graduate Faculty of Rutgers University.
The team used computational resources from the Princeton Institute for Computational Science and Engineering (PICSciE) and the Office of Information Technology (OIT) High Performance Computing Center and Visualization Laboratory at Princeton University for the in silico computer modeling.
It will be interesting to see of the models can work on the lab bench. Hydrogen has been in the doldrums of late like many other alternatives. The field needs a huge cost structure improvement.

