Jun
5
A New Process for Biofuel From Cellulose
June 5, 2012 | 3 Comments
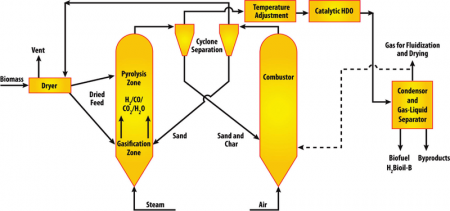
A new Purdue University-developed process for creating biofuels called H2Bioil heats biomass rapidly to about 500 degrees Celsius in the presence of pressurized hydrogen. Resulting gases are passed over catalysts, causing reactions that separate oxygen from carbon molecules and adding hydrogen (hydrodeoxygenation), making the carbon molecules high in energy content, similar to gasoline molecules.
The new Purdue developed biofuel process has shown potential to be cost-effective for production scale, opening the door for moving beyond the laboratory setting.
The economic analysis shows that the cost of the thermo-chemical H2Bioil method is competitive when crude oil is about $100 per barrel when using certain energy methods to create the hydrogen needed for the process. If a federal carbon tax were implemented, the biofuel would become even more economical.
The economic analysis was published in the June issue of Biomass Conversion and Biorefinery, showing that the energy source used to create hydrogen for the process makes all the difference when determining whether the biofuel is cost-effective. Hydrogen produced using natural gas or coal makes the H2Bioil cost-effective when crude oil is just over $100 per barrel. But hydrogen derived from other, more expensive, energy sources – nuclear, wind or solar – drive up the break-even point.
The conversion process was created in the lab of Rakesh Agrawal, Purdue’s Winthrop E. Stone Distinguished Professor of Chemical Engineering. Professor Agrawal said H2Bioil has significant advantages over traditional standalone methods used to create fuels from biomass, “The process is quite fast and converts entire biomass to liquid fuel. As a result, the yields are substantially higher. Once the process is fully developed, due to the use of external hydrogen, the yield is expected to be two to three times that of the current competing technologies.”
Wally Tyner, Purdue’s James and Lois Ackerman Professor of Agricultural Economics backs Agrawal up with, “We’re in the ballpark. In the past, I have said that for biofuels to be competitive, crude prices would need to be at about $120 per barrel. This process looks like it could be competitive when crude is even a little cheaper than that.”
Agrawal and his collaborators backed by the U.S. Department of Energy and the Air Force Office of Scientific Research have already received a U.S. patent for the conversion process.
The point of the economic analysis is the determining if the ‘glitch’ in all this is worth pursuing.
Working to develop catalysts needed for the H2Bioil conversion processes with Agrawal are colleagues Fabio Ribeiro, a Purdue professor of chemical engineering, and Nick Delgass, Purdue’s Maxine Spencer Nichols Professor of Chemical Engineering. The method’s initial implementation has worked on a laboratory scale and is being refined so it would become effective on a commercial scale.
Agrawal explains, “This economic analysis shows us that the process is viable on a commercial scale,” Agrawal said. “We can now go back to the lab and focus on refining and improving the process with confidence.”
The model Tyner used assumed that corn stover, switchgrass and miscanthus would be the primary feedstocks. The analysis also found that if a federal carbon tax were introduced, driving up the cost of coal and natural gas, more expensive methods for producing hydrogen would become competitive as well.
“If we had a carbon tax in the future, the break-even prices would be competitive even for nuclear,” Tyner said. “Wind and solar, not yet, but maybe down the road.”
So the new process is still lab bound, waiting for the catalysts that will reform the pyrolysis gasses into fuels. Seems we’ve looked at other efforts in the same position over the years. Pyrolysis is a great way to free the carbon out of biomass, its just the results, a lot of carbon dioxide, isn’t very cooperative in reacting into other products.
Perhaps the folks at West Lafayette could visit with the folks from Nottingham we looked at yesterday.
Biomass to fuel by pyrolysis will break out, it all hinges on the catalysts.
Comments
3 Comments so far


Why is it that biofuel people keep wanting the price of everything else to go up? (through taxes)
Why don’t they just concentrate on ways to take cost out of their own process.
Hey there would you mind sharing which blog platform you’re working with?
I’m going to start my own blog in the near future but I’m
having a tough time making a decision between BlogEngine/Wordpress/B2evolution and Drupal.
The reason I ask is because your design seems different then most
blogs and I’m looking for something completely unique.
P.S Apologies for being off-topic but I had to ask!
What’s up mates, its wonderful paragraph on the topic of culture and fully
defined, keep it up all the time.