Jul
8
A New Energy Storage Material
July 8, 2010 | 11 Comments
Washington State University scientists working at pressures found halfway to the center of the Earth created a new substance, which stores chemical energy in its very strong molecular bonds.
The team is saying the material could be used to create a new class of fuels or an energy storage device, although its practical application is still a long way off. That saying is getting lots of repeats across the Internet, too. The press release has great legs . . . on very thin disclosure.
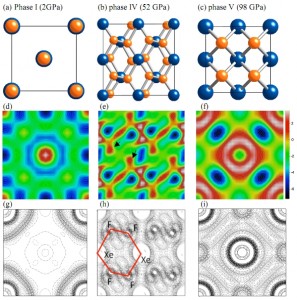
The scientists, led by WSU chemistry professor Choong-Shik Yoo, created the material by squeezing xenon difluoride (XeF2) – a white crystal used to etch silicon conductors – in a diamond anvil cell, a small, two-inch by three-inch-diameter device capable of producing extremely high pressures in a small space.
At normal atmospheric pressure, the material’s molecules stay relatively far apart from each other. But as the WSU scientists increased the pressure inside the chamber, the material became a two-dimensional graphite-like semiconductor. At around 50 GPa, the XeF2 transforms into a reddish two-dimensional graphite-like hexagonal layered structure of semiconducting XeF4. Above 70 GPa, it further transforms into a black three-dimensional fluorite-like structure of the first observed metallic XeF8 polyhedron.
This process transferred the device’s mechanical energy into chemical energy in the newly created molecular bonds.
Yoo told the UK website, The Engineer, “You can think of it as a polymer and the energy storage process was polymerizing the XeF2. About 90 per cent of the empty space in the substance was used to store energy.”
Yoo, explains all this as “squeezing” forced the molecules to make tightly bound three-dimensional metallic “network structures.” In the process, the huge amount of mechanical energy of compression was stored as chemical energy in the molecules’ bonds.
The research is basic science, but Yoo says it shows it is possible to store mechanical energy into the chemical energy of a material with such strong chemical bonds. Possible future applications include creating a new class of energetic materials or fuels, an energy storage device, super-oxidizing materials for destroying chemical and biological agents, and high-temperature superconductors.
The WSU scientists have created a compact, never-before-seen material capable of storing vast amounts of energy. Described by one of the researchers as “the most condensed form of energy storage outside of nuclear energy,” the material holds potential for creating a new class of energetic materials.
The amount of energy stored in the material has not yet been exactly calculated. Yoo is quoted by The Engineer saying it was comparable with a polymer form of nitrogen, which has three to five times the amount of energy found in the most powerful explosives used today.
“In the last 50 years, scientists have increased the power of explosives by about 10 per cent every decade. So a three to five times increase is enormous,” Yoo said.
The team is now studying ways of producing the material in larger quantities and ensuring its stability for use in practical applications.
This is very basic science. The uses noted above are in the realm of possibilities, not potential, nor can could, would or should enter the discussion. What’s missing is more obvious – does the new material keep its new volume when decompressed? Apparently so, the paper in Nature Chemistry has supplementary information with photographs, but the team is not discussing the material volumes at the described pressures.
The next item of interest is the comparison to nitrogen-based explosives. For some that might be a little unnerving, but the paper covers the material taking an electrical charge without blowing up. That’s not a conclusive safety test, but the XeF2 material isn’t an explosives material anyway.
Finally is the unnamed team member saying above, (the material is) “the most condensed form of energy storage outside of nuclear energy,” a very bold statement indeed. Without some kind of in depth explanation though, the comment seems more like hype or hyperbole.
But the door is opened. Forming up new materials under pressure is a new field. The WSU effort is showing that the compressing of molecules can affect their properties in desirable ways. All a lab basically needs is a diamond anvil and a suite of testing equipment with the necessary money.
Keep in mind, the WSU team has found a material that compresses, seemingly stays put and can be tested. There are surely lots more. Just where this new area in the field goes is anyone’s guess. Yet the idea of getting better or new use from the space between molecules and their atoms is an intriguing idea. Mass and volume can be difficult problems when thinking in the human scale for energy use.
Perhaps ultracompressed materials will yield worthwhile products sooner than we might think. If the comment above about the density is right, the field may well explode into furious activity very quickly.
Comments
11 Comments so far


It’s not clear how you would retrieve the energy – maybe they don’t know – but would it be heating or breaking the chemical bonds in some way?
Based on what I’ve read about EEStor, the miracle capacitor technology, they are doing something that is reminiscent of this. It appears that they are storing voltage potentials by using chemical bonds as well. If the theories about it are true, the storage is effective because the molecular bonds compress as a result of the gain in potentials, allowing it to store a lot of electricity and send it back out. Maybe I’m way off though.
[…] A New Energy Storage Material | New Energy and Fuel […]
I would like to exchange links with your site newenergyandfuel.com
Is this possible?
My partner and I really enjoyed reading this blog post, I was just itching to know do you trade featured posts? I am always trying to find someone to make trades with and merely thought I would ask.
Hey there! I’ve been following your website for a long time now and finally got the courage to go ahead and give you a shout out from Kingwood Texas! Just wanted to tell you keep up the great work!
Interesting read, perhaps the best article iv’e browse today. We learn everyday cheers to you!
Interesting read, perhaps the best article iv’e browse today. We learn everyday cheers to you!
I was just having a conversation over this I am glad I came across this it cleared some of the questions I had.
Hello, this is my first time i visit here. I found so many interesting in your blog especially on how to determine the topic. keep up the good work.
I’ve just started off a blog, the knowledge you give on this site has aided me extremely. Thank you for all your time & work.
I REALLY liked your post and blog! It took me a minute bit to find your site…but I bookmarked it. Would you mind if I posted a link back to your post?