Jan
19
A New Way to Make Methanol Fuel
January 19, 2010 | 6 Comments
University of Oxford researchers are developing a method for the homogeneous conversion of CO2 to methanol, with methanol as the only resulting C1 product. With momentum building in the methanol powered fuel cell market there is good reason to be on the lookout for the lowest cost and highest efficiency paths for producing methanol.
Andrew E. Ashley, Amber L. Thompson, and Dermot O’Hare at Oxford use a “frustrated Lewis Pair” (FLP)-based nonmetal-mediated procedure at only pressures of 1–2 atmospheres and 160 °C. There’s a head turner – very low pressure, and not particularly hot. The construction cost of such a plant and the energy used if the research makes it to commercialization would be much less than those using higher pressures and temperatures. 160 °C equates to only 320 °F and a 1-2 atmospheres is only 14.7 to 29.4 psi. This team is on to something; such operating conditions are very attractive.
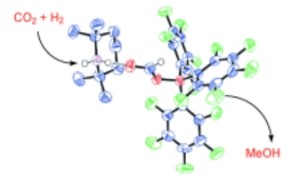
A frustrated Lewis pair (FLP) is a Lewis acid – base pair in which steric hindrance precludes the formation of a regular Lewis acid-base adduct. The steric hindrance occurs when the size of groups within a molecule prevent chemical reactions that would occur in related smaller molecules. Thus the “frustrated” effect. The Lewis acid-base adduct is a molecule formed by the direct addition of two or more distinct molecules causing a frustrated formation—as in ammonia borane. Kept from the “normal” reaction, these “frustrated” groups so turn to other molecules for reactions. That’s the opportunity the team has exploited.
As you’ll note if checking the (FLP) link that other research groups have shown that FLPs can activate H2 heterolytically, the cleaving a chemical bond effect. At first glance the hydrogenation of CO2 to the methanol molecule CH3OH might seem simple, but getting rid of the excess O and coming up with and attaching three H just right is a not so simple task. Current approaches to hydrogenation rely on solid oxide catalysts and are not exclusive to methanol output.
The team’s paper published in the December 21 2009 issue of the journal Angewandte Chemie International Edition points out in considering current approaches that, “. . . these systems tend to give mixtures of C1 products: CO, CH3OH, and CH4. Furthermore, we are not aware of the homogeneous conversion of CO2 into CH3OH with nonmetal complexes. . . Herein we describe the heterolytic activation of hydrogen and subsequent insertion of CO2 into a B-H bond in the first homogeneous process for the conversion of CO2 into methanol.”
The team used a FLP consisting of the base tetramethylpiperidine (TMP) and the acid B(C6F5)3, which had been shown in other work to cleave hydrogen (H2) to form the salt [TMPH] [HB(C6F5)3], “. . . upon the addition of CO2 (1 equiv) to a 1:1 mixture of TMP/B(C6F5)3 (4 equiv) in C7D8 under an H2 atmosphere, quantitative conversion into CH3OB(C6F5)2 via 2 was observed after 6 days at 160 °C. Remarkably, vacuum distillation of the solvent (100 °C) led to the isolation of CH3OH (17–25% yield based on integration of the 1H NMR spectrum against internal Cp2Fe and GC analysis) as the sole C1 product, alongside C6F5H and TMP by-products. We expect that the formation of methanol results from the reaction of CH3OB(C6F5)2 with TMP or its conjugate acid.”
The team is currently focusing investigation on increasing the stability of the system towards hydroxylic agents with the hope of thereby rendering the system catalytic.
What’s left out is what to do with the by-products and a needed recycling system. The increasing efficiency from 17 to 25% yield is an opportunity as well.
The possibilities are intriguing; these are mild conditions that can be created from concentrated solar and other low temperature sources and work below water faucet pressure. It might put a private “methanol economy” within reach if sources of hydrogen can be economically found to work at personal levels – and that’s the problem searching for low cost solutions.
Liquid fuel, a concentration of stored energy in the form of methanol is a very high potential path to personal energy storage. As methanol fuel cells improve and come down in price there’s going to be a market contest in fuels. One consumers can look forward to.
Comments
6 Comments so far


Mobil MeOH-to-Gasoline zeolite catalyst process is available for further processing. I
Interesting mechanism, must majorly tweak yield.
JP Straley
[…] A New Way to Make Methanol Fuel | New Energy and Fuel […]
Very interesting article, thank you.
You’ve got a great website at all
[…] the UK, researchers at Oxford university have developed a low-cost method to convert CO2 to methanol — using a “frustrated Lewis base-acid” in which a pair of molecules – that […]
Beneficial info and excellent design you got here! I want to thank you for sharing your ideas and putting the time into the stuff you publish! Great work!
thanks for your hard reseach and working on your project and is it possible to use this idea in car industies?
Thanks